Farzad Arjomandi Rad; Jila Talat Mehrabad
Abstract
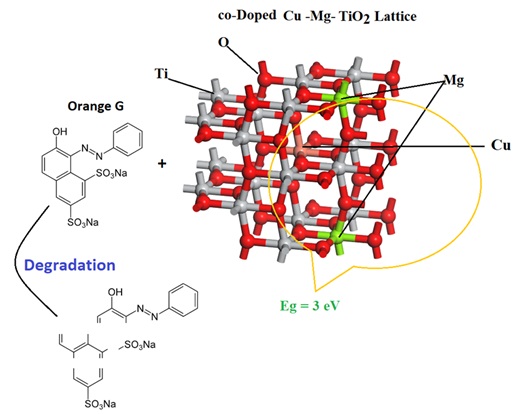
Pristine TiO2 nanoparticles, doped with single elements Cu and Mg, were synthesized utilizing the sol-gel process with titanium tetraisopropoxide as the Ti basis. The physicochemical ...
Read More
Pristine TiO2 nanoparticles, doped with single elements Cu and Mg, were synthesized utilizing the sol-gel process with titanium tetraisopropoxide as the Ti basis. The physicochemical characterizations of the nanoparticles were evaluated using X-ray diffraction (XRD), transmission electron microscopy (TEM), diffuse reflection spectroscopy (DRS), and scanning electron microscopy (SEM). The XRD shapes of the samples did not display isolated peaks of diffraction for Cu or Mg, demonstrating that the metals were well spread on the TiO2 surface. The DRS analysis uncovered an intriguing finding- the co-doped photocatalyst exhibited a significantly narrower band in comparison to both un-doped and monometallic TiO2. This intriguing alteration in the absorption band near visible light holds great potential. To further explore its implications, a comprehensive comparison was conducted to evaluate the photocatalytic activity of nanoparticles in degrading orange G solution under visible light. TiO2 nanoparticles doped with copper and magnesium exhibited significantly higher photocatalytic activity than to Cu/TiO2, Mg/TiO2, and pure TiO2 nanoparticles. The optimal doping levels of copper and magnesium for the synthesis of Cu and Mg/TiO2 nanoparticles were determined to be 1 and 0.25 mol%, respectively.