Document Type : Original Research Article
Authors
1 Department of Chemistry, Islamic Azad University, Ahvaz Branch, Ahvaz, Iran
2 Department of basic science, Farhangian University, Tehran 19989-63341, Iran
3 Department of Chemistry, Khuzestan Science and Research Branch, Islamic Azad University, Ahvaz, Iran
Abstract
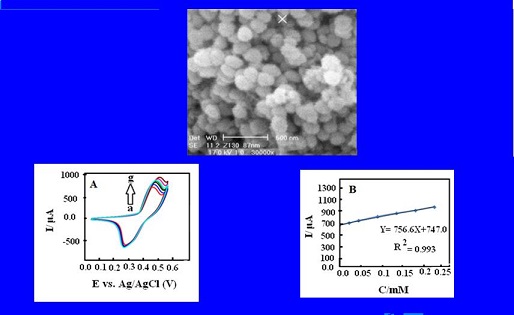
The aim of the present study was to determine the cefixime (CFX) through highly sensitive and simple electrocatalytic method. The electrocatalytic oxidation of CFX was performed on the surface of the modified carbon paste electrode (MCPE) with synthesized nano-sized ZSM-5 zeolite using the cyclic chronoamperometry and voltammetry methods. Also this work probed the application of the nano-zeolite in electrode structure and prepare zeolite MCPE. Due to the porous structure of the zeolite framework, the nickel (Ni) (II) ions were embedded into the zeolite framework through the immersing MCPE with synthesized zeolite in a 1.0 M Ni chloride solution. An excellent redox activity was practically seen for the Ni2+/ Ni3+ couple on the MCPE surface in alkaline solution. The Ni ions were acted as a mediator for the oxidation of CFX and catalyzed the electron transfer in this process. The CFX molecules were successfully oxidized on the surface of proposed electrode. The chronoamperometric method was used and catalytic reaction rate constant (K) was 3.5 ×106 cm3/s-1/mol-1 for the CFX oxidation. This electrocatalytic oxidation had a good linear response in the CFX concentration range of 25×10-6– 25×10−5 M with a regression correlation coefficient of 0.993, and the detection limit (3δ) of the method was 26×10-7 M. The diffusion coefficient of CFX molecules (D=6.47×10-5 cm2/s-1) was calculated based on the chronoamperometry studies.
Graphical Abstract
Keywords