Document Type : Original Research Article
Authors
1 Chemistry Department, Faculty of Science, University of Tripoli, Tripoli, Libya
2 Chemistry Department, Faculty of Science, Sebha University, Sebha, Libya
3 Department of Pharmaceutical Sciences, Faculty of Health Sciences, Sirte University, Sirte, Libya
Abstract
2-Thiohydantoin derivatives are most remarkably known because of their extensive applications including, hypolipidemic, anticarcinogenic, antimutagenic, antifungal, and antibacterial. For this reason, three of 5-aryl-2-thiohydantion derivatives (3-5) and the conversion of 2-thiohydantoin to thiol ether (7) by alkylation with methyl bromoacetate are successfully synthesized. To evaluate and understand the interactions and the orientation between the synthesized compounds and binding pocket of cancer and bacterial protein receptor, molecular docking studies using the Auto dock Vina were performed against three proteins including; isocitrate dehydrogenase (IDH1, PDB: 4UMX) and two bacterial proteins (LasR-OC12 HSL complex PDB:3IX3, and β-ketoacyl-acyl carrier protein synthase, PDB:1FJ4). The docking results demonstrated that lower free energy of binding (FEB) was obtained from the synthesized compounds ranging from −5.1 to −9.6 kcal/mol for 3IX3, −4.9 to −7.8 kcal/mol for 1FJ4, and −4.4 to −8.5 kcal/mol for 4UMX.
Graphical Abstract
Highlights
† These authors contributed equally to this work.
Keywords
Introduction
Thiohydantoins and their derivatives have been widely used in pharmaceutical purposes, including antimicrobial [1], antiviral [2], fungicides [3], antiparasitic [4], and anticancer [5]. They represent an important class of compounds within pharmaceutical chemistry and exist in various pharmacologically active molecules that possess significant bioactivities and are involved in therapeutic drugs. As an example for these important molecules, anticancer agent enzalutamide (MDV3100, ENZ), that used to treat castration-resistant prostate cancer (CRPC) [6]. Moreover, 5-substituted arylidene-2-thiohydantoin derivatives have been considered as potent inhibitors towards mutant isocitrate dehydrogenase cancer-associated enzyme IDH, the enzyme involved in the tricarboxylic acid cycle [7]. In addition, 5- arylidine-2-thiohydantoin derivatives have been reported as antibacterial agents that exhibited more than 90% inhibition of Mycobacterium tuberculosis growth [8, 9]. The 2-thiohydantoin derivatives containing triphenylamine moiety bounded through phenyl group linker in position C-5 and carboxymethyl substituent in N-3 position revealed high antibacterial activity with low cytotoxicity against human cells (U-937, HUT-78, and COLO-720L) [10]. 2-Thiohydantoins with substituted aryl groups at positions N1 and N3 showed potent antiparasitic activity against Trypanosoma brucei species [4]. Given the importance of these molecules, different methods have been used to prepare several of their derivatives. The most common approaches are based on the reaction of a-amino acids with i) acetic anhydride followed by treatment with ammonium thiocyanate, ii) isothiocyanate derivatives [11-13], iii) thiourea [14]. In addition, the reaction of N-methylthiourea with aldehydes to produce 2-thiohydantoin derivatives has been reported [15]. Another procedure to prepare 5-benzylidine 2-thiohydantion has been reported by the reaction of carbon disulfide followed by the addition of a primary amine to the product [16].
In general, these molecules contribute to drug discovery as they possess attractive features of their structures by owning a stereogenic center at position 5 as well as the feasibility of their synthesis. Therefore, the structure-activity relationship (SAR) of several thiohydantoin derivatives with different targets has been investigated [17]. In this regard, we aimed in this work to synthesize some 5-substitution 2-thiohydanition derivatives. In addition, we applied a molecular docking study using the Auto dock Vina program to investigate and understand the interactions and the orientations between the synthesized compounds and binding pocket of the target proteins’ receptors [18–27].
Experimental
General
All the chemicals were purchased from the Sigma-Aldrich, Fisher Scientific unless otherwise stated. Compounds are characterized by 1H NMR, 13C NMR experiments. NMR spectra were recorded on a Bruker Fourier 300 NMR spectrometer. Data are reported as chemical shifts in parts per million downfield from TMS, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet), coupling constant and assignment, respectively. IR spectra were recorded on a Perkin-Elmer 1600 series FTIR spectrometer. High resolution mass spectra were recorded by mass spectrometry. Melting points were recorded on a Gallenkamp MPD350 apparatus and are quoted uncorrected.
Synthesis of 3-Ethyl-2-thioxoimidazolidin-4-one (2)
To a solution of glycine (1.0 g, 13.3 mmol) in EtOH (2 mL) containing 1 equivalent of 50% aqueous KOH (2 mL) was added ethyl isothiocyanate (1.2 mL, 13.3 mmol) dropwise at 0 oC. The mixture was stirred for 3 h, and then acidified with 1 M HCl to precipitate the crude α-thioureido acid as a white solid at 0 oC, which was filtrated and dissolved in acetone and treated with H2SO4 at room temperature. The reaction completion was monitored by TLC, and the solvent was removed by reduced pressure, and saturated aqueous NaHCO3 was carefully added at 0 oC. The resulting precipitate was filtered as a white-yellowish solid (1.36 g, 71 %): Rf 0.66 (EtOAc:Hexane, 5:2). m.p. 151.3-153.7 oC. 1H NMR (300 MHz, CDCl3) δ 7.90 (s, 1H, NH), 4.09 (s, 2H, CH2), 3.88 (q, J= 7.1 Hz, 2H, CH2), 1.25 (t, J= 7.1 Hz, 3H, CH3). 13C NMR (75 MHz, CDCl3) δ 184.6 (C=S), 171.7 (C=O), 48.4 (CH2), 36.5 (CH2), 12.9 (CH3). HRMS (El): C5H8N2OS, Cal. 144.0400, found 144.0300.
General procedure for the synthesis of compounds (3,4,5 and 7)
A mixture of compounds 2 (200 mg, 1.39 mmol), sodium acetate (570 mg, 6.95 mmol, 5 eq.), and benzaldehyde (1.39 mmol) in 5 mL acetic acid was refluxed for 5 h. After the mixture was cooled, the formed crystals were washed successively with ethanol to afford the desired product.
(E)-5-Benzylidene-3-ethyl-2-thioxoimidazo- lidin-4-one (3)
Yellow crystals (207 mg, 68%, Rf 0.91; EtOAc:Hexane, 5:2), m.p. 178.7-180.0 oC. 1H NMR (300 MHz, CDCl3) δ 9.15 (s, 1H, NH), 7.67-7.31 (m, 5H, Ar-H), 6.76 (s, 1H, CH=Ph), 3.97(q, J= 7.2 Hz, 2H, CH2), 1.30 (t, J= 7.1 Hz, 3H, CH3). 13C NMR (75 MHz, CDCl3) δ 178.1 (C=S), 163.6 (C=O), 132.8, 129.8, 129.5, 129.2, 126.5 (Ar-C), 113.5 (CH=Ph), 36.7 (CH2), 13.0 (CH3). MS(EI): C12H14N2S, calculated 218.0900, Found 218.0664. u cm-1: 3352.3, 2966.5, 1708.9, 1643.4, 1460.1, 1404.2, 763.8, 682.8, 542.0, 457.1.
(E)-3-Ethyl-5-(4-hydroxybenzylidene)-2-thioxoimidazolidin-4-one (4)
Yellow-brownish crystals (213 mg, 62%, Rf 0.73; EtOAc:Hexane, 5:2), m.p. 193.2-195.5 oC.1H NMR (300 MHz, CDCl3) δ 9.89 (s, 1H, OH), 7.83 (d, J= 8.1 Hz, 1H, 2 CH-Ar), 7.28 (s, 1H, CH=Ph), 7.11 (s, 1H, NH), 6.98 (d, J= 8.2 Hz, 2H, 2 CH-Ar), 3.90 (q, J= 7.1 Hz, 2H, CH2), 1.72 (s, 1H, NH), 1.27 (t, J= 7.1 Hz, 3H, CH3). 13C NMR (75 MHz, CDCl3) δ 163.8 (C=S), 161.5 (C=O), 132.5, 130.1, 129.6, 129.3, 128.7 (Ar-C), 116.5 (CH=Ph), 42.8 (CH2), 12.9 (CH3). MS(EI): C12H14N2S, calculated 248.0600, Found 248.0620. u cm-1: 3371.6, 1707.0, 1643.4, 1581.6, 1240.2, 1165.0, 825.5, 644.2, 484.1.
(E)-2((1-Ethyl-5-oxo-2-thioxoamida zolidin-4-ylidine) methyl)- benzoic acid (5)
Yellow crystals (190 mg, 52%), Rf 0.88 (EtOAc:Hexane, 5:2), m.p. 225.7-227.1 oC. 1H NMR (300 MHz, CDCl3) δ 8.61 (s, 1H, NH), 7.87-7.65 (m, 3H, CH-Ar), 7.34 (s, 1H, CH=Ar), 3.95 (q, J= 7.1 Hz, 2H, CH2), 1.34 (t, J= 7.1 Hz, 3H, CH3). 13C NMR (75 MHz, CDCl3) δ 160.7 (C=S), 158.0 (C=O), 134.3 (C=O), 133.1, 130.9, 130.1, 129.6, 129.5 (Ar-C), 127.1 (CH=Ph), 36.4 (CH2), 12.6 (CH3). MS(EI): C13H14N2O2S, calculated 262.0800, Found 262.9882. u cm-1: 3082.2, 2972.3, 1726.3, 1685.8, 1352.1, 1265.3, 1074.4, 759.9, 684.7, 621.1.
Methyl 2-((1-ethyl-5-oxo-4,5-dihydro-1H-imidazol-2-yl)thio)acetate (7)
To a solution of 2 (100 mg, 0.69 mmol) in acetone (20 mL) was added powder K2CO3 (100 mg, 1.38 mmol) and methyl α-bromoacetate (1.18 eq. 75 µl, 0.81 mmol). The reaction mixture was stirred at r.t for 24 h. The precipitate was removed by filtration, and the filtrate was concentrated and then purified by flash chromatography (CH3Cl:MeOH; 10:1, Rf 0.91) to give 10 as brown oil (71.1 mg, 47%). 1H NMR (300 MHz, CDCl3) δ 4.37 (s, 2H, CH2), 4.04 (s, 2H, CH2), 3.71 (s, 3H, CH3CO), 3.57 (q, J= 7.2 Hz, 2H, CH2), 1.27 (t, J= 7.2 Hz, 3H, CH3). 13C NMR (75 MHz, CDCl3) δ 184.3 (C=O), 179.6 (C=O), 161.9 (C=N), 59.2 (CH2-S), 53.5 (OCH3), 37.5 (CH2CO), 32.8 (CH2), 19.1 (CH3). MS (EI): Calculated 216.0600, Found 216.0525.
Results and Discussion
Chemistry
The synthesis of 2-thiohydantoin 2 was carried out following the reactions, as shown in Scheme 1, where glycine 1 is treated with ethyl isothiocyanate in ethanolic potassium hydroxide followed by neutralization with sulfuric acid. Condensation of 2-thiohydantion 2 with aromatic aldehydes (benzaldehyde p-hydroxybenzaldehyde and 2-carboxybenzaldehyde) was performed in the presence of sodium acetate in acetic acid under reflux, affording 3, 4 and, 5 respectively. However, aliphatic aldehyde (cyclohexanecarboxaldehyde) failed to react with compound 2 to produce the condensation product 6 despite using different conditions such as using higher concentrations from sodium acetate and increase the reaction time and temperature (Scheme 1). To prepare biologically active 2-aminoimidazole-4-one derivatives 8, compound 2 was treated with methanolic ammonia [28]. but unfortunately, this reaction was not successful. Whereas, alkylation of 2-thiohydation 2 by methyl bromoacetate provided compound 7 [29], which can be used in further reactions (Scheme 1).
Scheme 1. Reactions of 2-thiohydantion (2) with different aldehydes (I, ii, iii, and iv) in the presence of AcONa/AcOH. i) Benzaldehyde. ii) 4-Hydroxybenzaldehyde. iii) 2-Carboxybenzaldehyde. iv) Cyclohexanecarboxaldehyde. v) BrCH2COOMe/K2CO3, Acetone. vi) NH3-CH3OH, r.t.
Molecular docking
Receptors X-ray Structures
A molecular docking study was carried out using the Auto dock Vina program [30] to investigate the binding interaction modes and affinities between five synthesized compounds (2, 3, 4, 5, and 7) and the binding pocket of the target proteins. The compounds were sketched using Chem Draw 12 software. Hydrogen bonds were added, and geometry optimization is performed using hperchem software. Racoon software was used to convert the synthesized ligand to PDB coordinate files to be ready for the molecular docking procedure. The crystal structures of the used proteins were obtained from the Protein Data Bank (PDB: 1FJ4, 3IX3, 4UMX) [31, 32]. Auto Dock Tools (ADT) was used to edit the proteins by the removal of unwanted water molecules and hetero atoms and then polar hydrogens were added followed by computing Gasteiger and adding Kollman charge. Finally, proteins were saved as pdbqt format. Discovery studio visualizer 2016 was utilized to identify the binding sites. It shows the amino acids that form the binding sites as binding to the coordinated ligand. Our compounds interacted with the same amino acids and other amino acids on the protein. For the protein with PDB: 1FJ4, the binding site consists of amino acids as follows Gly305, Pro272, Thr302, Phe390, Ala271, Thr300, val270, Phe392, Phe229, Gly391, His298 Thr115, Asp73, Gly205, Thr203, Pro303, Val304, Ala602, Val720, for the protein with PDB: 3IX3 the amino acids in the binding site are Thr115, Asp73 Trp88 Ala127, Tyr47, Ala50, Leu36 Tyr56, Tyr64, Gly268, Asp279, Ser129, Val76, Ala105, Leu110, Thr75, Tyr93, Phe101, Trp160, Arg61 Ser120, and for the protein with the PDB: 4UMX consists of amino acids as follows Gly268, Asp279 Ser287, Leu 288, Met290, Met291, Pro118, Leu120 Arg119, Ala111, Ile128, Trp124, Ile130 Ala282, Val280 Tys285, Val281, Gly286, Ile112, Ile117, Phe390, Phe392, Phe229, Val720, Ala271, Cys114, Ile113. During the docking process, the center grid parameter was specified for x, y and z-axis as of 60-56-60, respectively, with a spacing of 0.375Ao (Figure 1).
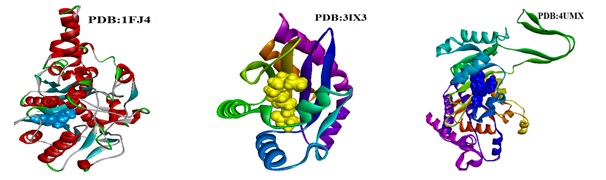
Figure 1. A schematic representation of the 3D structure of β-Ketoacyl-Acyl Carrier Protein Synthase (1FJ4) coordinated with thiolactomycin red color in CPK format. A Schematic representation of the 3D structure of LasR-OC12 HSL complex (3IX3) coordinated with PHSRRHGYXQCRPU-AWEZNQCLSA-N blue color in CPK format. IDH1 R132H in complex with cpd 1
Control docking
Before performing the molecular docking between the synthesized compounds and the target proteins, the validation of the docking protocol was evaluated to be re-docking the coordinated ligand with the proteins (PDB: 4UMX, 1FJ4, and 3IX3). It has already been confirmed that the significance of the binding affinity of standard drugs is approximately 6.0 kcal/mol or higher [33-36]. The docking results showed that the binding conformation of re-docked the coordinate was found to be identical to the experimental binding position inside the binding pocket with the essential amino acids. These results indicate that the docking software can reproduce the expected binding mode of the co-crystallized ligand. Besides, the results of validation showed the binding affinity -7.1 kcal/mol, -7.2 kcal/mol, and -7.8 kcal/mol. The obtained results can be trusted and applied for the test compounds with the target protein when the RMSD value obtained is less than 2 [37, 38]. Furthermore, the RMSD can be used to determine the degree of deviation between the experimental and crystallographic ligands in the same binding pocket. The deviation was higher when the RMSD value was high, indicating a high prediction error of the protein-ligand interactions [39]. On the other hand, the smaller RMSD value obtained reveals better conformation because the redocking ligand position is closer to the crystallographic ligand position [40]. The RMSD value of the three crystallographic ligands was 1.47A, 1.00 A. 2.00 A for 4UMX, IFJ4 and 3IX3, respectively, indicating that the method employed for the docking procedure is valid and can be applied to other ligands with the same binding site region as shown in Figure 2.
Figure 2. The Superimposition between the docked conformation (yellow), and the crystal structure (orange and green) with 1FJ4, docked conformation (green), and the crystal structure (yellow) for 3IX3 and docked conformation (yellow), and the crystal structure (green) for 4UMX
The obtained results showed that three compounds including, (E)-5-Benzylidene-3-ethyl-2-thioxoimidazolidin-4-one (3), (E)-3-Ethyl-5-(4- hydroxybenzylidene)-2-thioxoimidazolidin-4-one (4), and (E)-2((1-Ethyl-5-oxo-2-thioxoamidazolidin-4-ylidine)methyl)benzoic acid (5) exhibited Low binding energy against the three used proteins, while the other two compounds did not display any inhibition against the same three proteins. The possible reason for this could be due to the absence of phenyl ring and any free function group such as hydroxy or carboxyl groups in comparison with the other three active compounds (Table 1). The resulted minimum FEB is ranging from −9.6 to −5.1 kcal/mol for 3IX3, − 7.8 to −4.9 kcal/mol for 1FJ4 and −8.5 to −4.4 kcal/mol for 4UMX (Table 1).
Generally, a protein-ligand complex that shows the lowest FEB is considered to be a potential inhibitor [41–45]. According to the results presented in Table 2, the three compounds that exhibited the lowest FEB (5,4 and 3) were found to be the potential candidates. The interactions between the docked compounds and the target proteins were manually examined. Extensive interactions can be seen between the synthesized compounds and the essential residues in the binding pocket. These interactions include hydrogen bonding, Van der Waals, Alkyl, pi-pi T-shaped, Hydrophobic (not showed), and electrostatic interactions.
Table 1. Results of Auto dock Vina for the five synthesized compounds
By performing molecular docking, reliable conformations were obtained for the synthesized compounds at the used enzymes binding pocket, and the results presented the functional and structural insight into the nature of the interactions. The molecular docking of the three synthesized compounds (3, 4, and 5) against the protein 4UMX showed different interactions similar to those with the coordinated drug. Compound 5 formed H-bond with Gly268, Asp279, Van der Waals with Ser287, Leu 288, Met290, Met291, Pro118, Leu120, Alkyl with Arg119, Ala111, Ile128, Trp124, Ile130Ala282, Val280 and Pi-Pi-T-shaped interaction with Tys285. While compound 4 displayed Van der Waals interaction with Val281, Gly286, Ile112, Ser287, Met290, Ile117, Pro118, Met29, Alkyl with Ile130, Ile128, Leu120, Arg119, Ala111and Pi-Pi-T-shaped with Tyr285, Trp124. Similarly, the exhibited interactions in compound 3 were H-bonding with amino acids Ser287, Van der Waals with Trp124, Met291, Gly286, Ile117, Cys114, Ile112, Alkyl with Val281, Leu120, Arg119, Ile113, Ala111, Leu120, Pi-Pi-T-shaped with Tyr285 and Carbon H-bond with Pro118 (Table 2 and Figure 3).
The docking results of the molecules 5 with the protein 1FJ4 with the essential amino acids in the binding pocket 8 displayed H- bonding with Gly305, and another interactions carbon H-bond with Pro272, Thr302, as well as Van der Waals interactions with Phe390, Ala271, Thr300, val270, Phe392, Phe229, Gly391, finally Pi-cation interaction with His298. Compound 4 exhibited H-bonding with Gly205, Gly305, as well as Van der Waals with Thr203, Thr300, Phe390, Phe392, and alkyl interactions with Pro303, Val304, Ala602, Pro272. Compound 3 formed Van der Waals interactions with Phe390, Phe392, Phe229, Val720, Ala271, as well as Alkyl with amnio acids Val304, Pro303, Ala602 as well as carbon H-bond with Pro272, Thr302 (Table 2 and Figure 3). The synthesized compound exhibited an interaction with 3IX3 at the binding pocket. Compound 5 showed H-bond with important residues Thr115, Asp73, also Van der Waals interaction formed with Trp88, Alkyl interaction with Ala127, Tyr47, Ala50, Leu36, as well as Pi-Pi-T-shaped interaction with reside in Tyr56, Tyr64, and Pi-anion with Asp73.
Table 2. Binding interactions details of the 3,4 and 5 compounds docked at the binding pocket of the target proteins 1FJ4, 3IX3, and 4UMX
Compound 4 demonstrated carbon H-bond with Trp88, Ser129, alkyl interaction with Ala127, Val76, Leu36, Ala105, Leu110, and Pi-Pi-T-shaped with Tyr64, Tyr56. Compound 3 exhibited Van der Waals with restudies Thr115, Thr75, Tyr93, Phe101, Trp160, Arg 61, as well as Carbon H-bond with Ser120, alkyl interaction with Leu110, Ala105, Leu36, Val75, Ala127, Pi-anion also formed with Asp73, Pi-Pi T-shaped with Tyr285, finally Carbon H-bond formed with Pro118 (Table 2 and Figure 3). As it can be noted from the results above, the three compounds 3,4, and 5 exhibited high binding affinity and good binding interactions with the essential amino acids in the binding pockets (Figure 4) with the important amino acids residues similar to those interactions are shown by the coordinate ligands, which make our compounds consider as a potential candidate for isocitrate dehydrogenase cancer-associated enzyme (IDH1), and as an antibacterial agent for Escherichia coli [46] and Pseudomonas aeruginosa [47].
Figure 3. Two-dimensional (2D) binding modes of the three compounds (3,4, and 5) present at the binding pockets of the target proteins 4UMX, 1FJ4, and 3IX3
Figure 4. The Superimposition between the docked conformation of the three synthesized compounds 3,4, and 5 in the binding pockets of the target protein. A 4umx, B 3IX3, and C 1FJ4
Conclusion
In this research study, 2-thiohydantoin (2) and its three 5-aryl-2-thiohydation derivatives, as well as a new thiol ester were synthesized following reported procedures, and their structures were characterized by spectroscopy techniques. In silico molecular docking studies for these synthesized compounds were successfully applied against three proteins as anticancer and antibacterial agents. These compounds exhibited different interaction modes with the essential amino acids in the enzymes binding pockets and displayed lower binding energy comparing to the coordinate ligand. The obtained results helped to identified potent new anticancer and antibacterial agents. According to the molecular study and the previous studies, we expect that the synthesized compounds will display potent anticancer and antibacterial activity particularly the molecule that contains carboxylic group on the phenyl ring. However, experimental studies in vitro are needed to confirm this finding.
Acknowledgment
The authors would like to express their sincere appreciation to their universities, Tripoli, Sebha and Sirte, for providing the resources and services.
Conflict of interest
The authors declare no conflict of interest.




.jpg)




