Document Type : Review Article
Authors
- Emmanuel Ibukun Aduloju 1, 2
- Noorfatimah Yahaya 2
- Nadhirah Mohammad Zain 2
- Mohammad Anuar Kamaruddin 2
- Muhammed Ariffuddin Abd Hamid 2
1 Department of Science Technology, School of Applied Science and Technology, The Federal Polytechnic Offa, P.M.B. 420, Kwara State. Nigeria
2 Department of Toxicology, Advanced Medical and Dental Institute (AMDI), Universiti Sains Malaysia, 13200 Bertam Kepala Batas, Penang, Malaysia
Abstract
The extraction process of bioactive components from nutraceutical, food, cosmetic industries, and pharmaceutical is a crucial task in the manufacturing process of natural products that demand green solvents. This all-important process needs the inclusion and utilization of natural deep eutectic solvents (NADES) to yield green products safe for the consumption of human beings and eco-friendly. This value-added green solvent has caught the attention of many researchers worldwide, cutting across different fields owing to its unique properties and vast potential. Also, the ability of the novel NADES to replace ionic liquids and conventional organic solvents in the extraction, and most especially in that of bioactive media, makes it an outstanding feat because it complies with the protocol for green analytical chemistry. In addition, the hydrophobic DESs (HDES) are also considered a suitable category of DES that needs to be explored in the field of extractions because of their stability in contact with water, a problem with using other classes of DESs. The HDESs possess exceptional tunable properties suitable for the optimum extraction and isolation of bioactive compounds, volatile fatty acids, and heavy metals from an aqueous medium. This paper explores and extensively discusses in review the prowess, and properties originating from the characteristics and functionalities of NADES and HDES, harnessing the solubility, other physicochemical characteristics, and possible interactions depending on the prevailing environmental conditions towards the extraction of these compounds.
Graphical Abstract
Keywords
- Extraction method
- Bioactive compounds
- Natural deep eutectic solvents
- Natural products
- Green chemistry
- Ionic liquids
- Nutraceutical
Main Subjects
Introduction
In the medieval era, the Romanians and Grecians utilized a lot of medicinal herbal plants and extracts, such as coriander and castor oil, for therapeutic uses, as reported by many researchers like Hippocrates, Celsus, Dioscorides, and Theophrastus. They utilized bioactive components from natural flora in ancient times for diverse prospects such as medicinal therapeutic, cosmetics, preservative, and nutritional purposes, thus improving the human community's health status during diseases [1]. Bioactive compounds are naturally occurring components originating from microbes, plants, fungi, fruits, and algae, requiring different isolation and extraction approaches. Bioactive compounds can be divided into two broad areas; Firstly, the primary metabolites are chemical compounds such as lipids, proteins, carbohydrates, and amino acids that aid growth and development in the body. Secondly, the secondary metabolite originates from plants and is believed to combat deleterious and infectious diseases [2]. These secondary metabolites are initially produced by the plant as a defensive and stress-response mechanism to combat toxic pathogens, predators, and herbivorous animals and survive the overwhelming challenges in their immediate environment [3]. Also, they can be synthesized by floral species inform of aroma to attract insects for their fertilization and pollination. Such natural products have been reported to have significant benefits as supplements in the human body and exhibit arrays of analgesic, antimicrobial, antidiarrheal, anti-inflammation, anticancer, antiaging, and antioxidant properties. According to World Health Organization, about 80% of the world’s population depends on natural products and medicines. Also, about 60% of approved anti-cancer pills emanate from natural bioactive compounds such as phenylpropanoids, polyketides, alkaloids, isoprenoids, and flavonoids. These bioactive compounds also contain prominent natural phytochemicals needed in the human body, such as fat-soluble vitamins E (tocopherol), Ascorbic acid (C), and vitamin A (beta carotene) alongside polyphenols. The essential and novel therapeutical properties of bioactive compounds have garnered the attention of researchers, especially in pharmaceutical fields, to use them in placesenstead of synthetic compounds [4].
Also, researchers have developed and imbibed a green effective extraction mechanism for bioactive compounds since their growing demand in large quantities for agricultural, industrial, nutraceutical, pharmaceutical, cosmetics, and even food industries. Their urgent need has also been greeted with the quest for eco-friendly extraction and recovery approaches, especially considering extraction techniques and the suitable solvent needed [4].
The sole aim of the extraction, synthesis, and sample preparation approach with functional material is to reduce and replace the application of deleterious chemical, costly and toxic materials, consume less energy, reduce overall reaction time, minimize solvent waste in the sample preparation approach and enhancing, ensuring safety by providing environmentally responsible alternatives during chemical analysis, thus making the entire analytical process green [4]. Green sample preparation techniques used in this process include microwave-assisted extraction (MAE)[5-7], solvent extraction, pressurized liquid extraction (PLE) [8,9], ultrasound-assisted extraction (UAE) [10,11], QuEChERS (Quick, Easy, Cheap, Effective, Rugged and Safe), supercritical fluid extraction (SFE)[12], and ionic liquids (IL)[13,14]. These technologies show speed, fewer steps, and greater yields than the traditional extraction methods of bioactive compounds from complex environmental matrices. Selecting a suitable solvent is essential to any of the extraction techniques used in the extraction and recovery of bioactive compounds and any other target analytes. This is due to diverse bioactive constituents exhibiting different physicochemical properties and polarities originating from their functionalities, thus affecting their solubility and dispersion in any medium.
Deep Eutectic Solvent
Generally, conventional organic and petroleum-based solvents have been exploited over the years for solvent extraction because of the good extraction potential exhibited towards polar and non-polar compounds based on polarity. However this has some demerits such as toxicity, non-biodegradable, non -biocompatible, high flammability, carcinogenicity, corrosivity, and high volatility [15]. Examples include acetone, methanol, ethyl acetate, and their mixtures. Even though these ionic liquids (IL) have their disadvantages, as earlier stated, the emergence, development, and introduction of new class solvents called the Deep Eutectic Solvents (DESs) that are economical, eco-friendly, highly- biocompatible, non-toxic, biodegradable with adjustable viscosity and simple in fabrication and much better when compared to the ILs have evolved [16]. The word “Eutectic” comes from the Greek "ευ" (eu means easy), and "Τήξις" (this means melting) is described as a mixture of a compound capable of forming a joint super lattice that can melt and simultaneously freezes at temperature (60 °C) lower than the melting points of their separate individual components. DES is synthesized from solid compounds, which become liquid when in proper molar ratio and at a specific combination. There are three different approaches to mixing DESs: (a) Thermal mixing on the hot plate at 80°C with vigorous continuous mixing using a magnetic stirrer for less than 2 h producing a clear colorless liquid. (b) Complete dispersal and dissolution in water prior to vacuum evaporation in a rotatory evaporator at 50oC and finally, (c) freeze-drying dissolved components in an aqueous medium.
Types of DESs
DESs are synthesized and represented based on the complex components used and are subdivided into five types, as shown in Figure 1: Type I The general formula is Cat+X−zMClx where X− is a Lewis base (x and z refer to the number of Cl− and MClx, respectively). Non-hydrated metal halides make up the bulk of DESs. (MClx) and quaternary ammonium salt (Cat+X−). The low melting point requires a small number of non-hydrated metal halides.
Type II Hydrated metal halides make up this class of DESs. (MClx.yH2O) and salts (y is the number of H2O molecules).
Type III combination of two or more of the following: choline chloride (ChCl) with HBDs like alcohols, amides, and carboxylic acids.
Type IV these DESs are formed from the mixture of HBDs with suitable metal halides. For example, Abbott et al. [16] formed eutectic mixtures and reported the combination of ZnCl2 with various appropriate HBDs, including ethylene glycol, urea, acetamide, and 1,6-hexandiol. finally,
Type V this type of DESs is formed from the mixtures of non-ionic compounds to synthesize a new class of DES with decreased freezing points [17].
 Figure 1. Subdivisions of DESs
Figure 1. Subdivisions of DESs
In contrast to hydrophobic DES, which limited because of the low number of synthesized hydrophobic DESs arising from limited number of available cheap hydrophobic salts and other constituents that yield eutectic mixtures at < 50oC. The interactions between the HBA and HBD existed due to the hydrogen bond originating from the HBA, and their hydrophobicity is attested to by low WC (water content) < 1.8% and low salt leakage value < 1.9% upon interaction with water. As reported by various researchers, the HDES can be categorized into neutral-based and salt or ionic-based DES and is effective in extracting volatile fatty acids from aqueous medium [18-20]. Examples of HDESs are tetraoctylammonium bromide, methyltrioctylammonium chloride, methyltrioctylammonium bromide, methyltrioctylammonium chloride, and tetraheptylammonium chloride. Other types of HDES include combinations of two neutral compounds like natural Hydrophobic HBAs such as L-menthol, terpenoids, and thymol [21] combined with HBDs like alcohols with long-chain acids and carboxylic acids. In another report, other neutral-based HDESs are synthesized from combinations of fatty acids acting as both HBAs and HDBs, such as lauric acid, lidocaine combined with either decanoic oronanoic or octanoic acid [22].
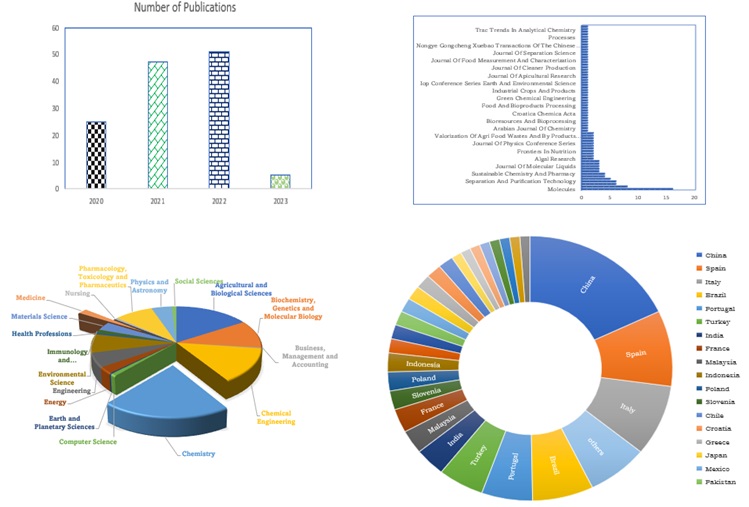 Figure 2. (a) Growing Numbers of Journal publications. b. Journals Sources with NADES extraction of bioactive compounds (c) subject areas of researchers whose journals reviewed (d) Countries where publications concerning NADES applications using the keywords “natural deep eutectic solvents for solvent extraction of bioactive compounds” (source www.scopus.com; date of search: 26.01.23).
Figure 2. (a) Growing Numbers of Journal publications. b. Journals Sources with NADES extraction of bioactive compounds (c) subject areas of researchers whose journals reviewed (d) Countries where publications concerning NADES applications using the keywords “natural deep eutectic solvents for solvent extraction of bioactive compounds” (source www.scopus.com; date of search: 26.01.23).
In this review, 128 publications were on natural deep eutectic solvents applications from across the world's continents, asshown in Figure 2 a-d. The major path of which are from Asia and Europe, while others, like North and South America, are works in progress. These designer solvents have been applied for various purposes because of their tunable Physicochemical properties, diverse structural potentials, and characteristic interactions with specified analytes, trace, and ultra-trace compounds, thus enhancing their selective extraction and recovery from their complex matrices [23]. Other researchers have reported and published different views on the green methods of extracting and isolating plant biologically active compounds and HDES, natural products using NADES, subcritical water, and supercritical fluid for their extraction. Also, the study and number of publications credited to HDES is still in their infancy and steadily increasing from 2015-2023 across various fields such as bioengineering, biotechnology, biomedicine, nanotechnology, separation, and extraction technology.
This can be inferred from the increasing number of research publications between 2020 to 2023, contributing to the potential applications and biological activities, while some emphasized the chemical interactions and vital experimental factors that aided the extractions [24-26]. Special Emphasis has also been placed on NADES potential to effectively extract carrageenans, α-mangostin, flavonoids, tools, apigenin, genistin, isoflavones, keratin, xanthones, tanshinone, and phenolic acids from complex food, biological and environmental matrices and the suitable microextraction techniques such as LPME, UAME, and MAE. This review summarizes the structure, formation, and physicochemical properties of natural deep eutectic solvents (NADES) and HDES to extract and recover bioactive compounds from plants and food sample complexes. This interesting study also shed more light on their use in chromatographic and extraction media concerning parameters like biocompatibility, solubilizing, biodegradability, conductivity, hydrophilicity, polarity, renewability, and stabilizing abilities.
Types and synthesis of selected bioactive compounds
Bioactive compounds are categorized into three (3), namely: alkaloids, phenolic compounds, and terpenes
Alkaloids
The term “alkaloid” was derived from the Arabic name al-qali, a plant from which soda was first extracted. It was first introduced by a renowned German researcher Carl F. W. Meissner, years ago. These consist of nitrogen-containing cyclic organic compounds naturally found in fungi and bacteria, but most especially in plants. They are biosynthetically made from purine and amino acids. They consist of an aliphatic or aromatic heterocyclic ring with a nitrogen atom with a lone paired electron. They also exist in diverse chemical structures based on their bioactivity and defense mechanisms against extreme, dangerous environmental situations inform of bitter taste and toxicity. They are known for their anti-inflammatory, anesthetics, and anesthetics agents such as nicotine, ephedrine, strychnine, nicotine, and morphine. Also, like the imidazole alkaloids, they exhibit cytotoxic activity against human ovarian carcinoma, tumor cell lines, human pancreatic adenocarcinoma, and human bladder carcinoma [27]. Alkaloids are prevalent in Boraginaceae, Loganiaceae, Leguminoceae, Solanaceae, Berberidaceae, Annonaceae, Apocynaceae, Piperaceae, and Rubiaceae. They are classified based on origins or their derived biological pathways, molecular precursor, and structure. Generally, alkaloids are classified as true alkaloids, protoalkaloids, and pseudoalkaloids, except for pseudoalkaloids; others are prepared from amino acids.
True Alkaloids
These alkaloids are derived from amino acids. They consist of an aliphatic or aromatic heterocyclic ring characterized by nitrogen atoms having lone paired electrons. They are crystalline, highly reactive, potent in biological activities, form water-soluble salts, and conjugate with acid to form a salt. Apart from nicotine, a brownish liquid, this class of alkaloids is solid and bitter. They occur naturally in plants as salt, Free-state, and N-oxide. Precursors of this group are l-ornithine, l-phenylalanine/l-tyrosine, l-lysine, and l-histidine. Common Natural forms
Protoalkaloids
They are derived from amino acids with nitrogen atoms having lone paired electrons but not an aromatic heterocyclic ring system. The main precursors of this group are l-tyrosine and l-Tryptophan, forming mescaline, yohimbine, and hordenine. They are used as anesthetics for various health disorders such as pain, mental illness, and neuralgia.
Pseudo Alkaloids
They are derived from amination pathways and transamination reactions from the post-cursors of amino acids, phenylalanine or acetate, and non-amino-acid precursors. Examples of alkaloids in this group are caffeine, capsaicin, and ephedrine. Other alkaloids classified based on the availability of basic heterocyclic nuclei in their structure are Pyrrolidine alkaloids, Purine alkaloids, Imidazole alkaloids, Steroidal alkaloids, Indole alkaloids, Isoquinoline alkaloids Quinolines alkaloid, Pyrrolizidine alkaloids, Piperidine alkaloids, and Tropane alkaloid.
Phenolic Compounds
These antioxidants consist of flavonoids, tannins, lignins, phenol, and phenolic acid with carboxylic acid, hydroxybenzoic and Hydroxycinnamic acids in their chemical structure. Generally, phenolic compounds are classified intohenolic acids and polyphenols [28]. The major phenolic phytochemicals isolated from plants, especially fruits and vegetables, are hydroxycinnamic acids such as p-coumaric caffeic acid, sinapic, ferulic, and hydroxybenzoic acids such as ellagic acid and gallic [29]. These phenolic compounds are antioxidants specializing in cell wall stability and are deposited on the outer layers of the backs and skins of plants. They have also been proven to possess antitumor, antibacterial and anti-inflammatory properties.
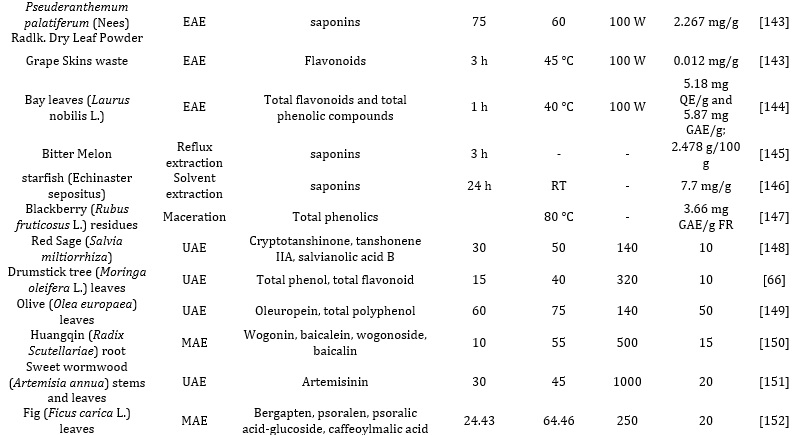
Extraction solvents have been applied to extract different bioactive compounds such as alkaloids, terpenoids, phenolic compounds, and polysaccharides [30]. These phenolic functionalities include lignan, flavonoids, phenolic alcohol, and phenolic acids. In order to overcome the shortcomings experienced while extracting this phenolic compounds via the Soxhlet and the organic solvent-based techniques, greener extraction approaches such as Ultrasound- assisted approaches (UAE), Supercritical Fluid Extraction Approaches, and microwave-assisted extraction (MAE) have emerged and applied [31,32] as summarized in table 1 and 2.
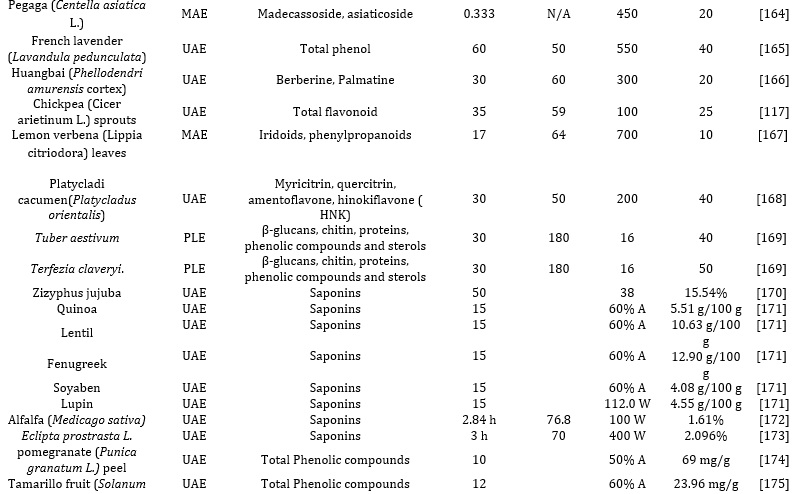
Thus researchers have employed different multiple extraction methods to enhance the yield of phenolic compounds, includinghe use of DESs and organic acid-based DESs as the extraction solvent [33]. Researchers have developed and implemented an optimized method of extraction for chlorogenic acids from environmental matrices such as cocoa and coffee, using the novel DES over conventional organic solvents [34,35]; the designer ChCl: lactic acid-based DES(molar ratio 1:2) employed yielded excellent and satisfactory results making it a good alternative when compared with traditionally organic solvents. Meanwhile, the polyalcohol-based DESs did not show satisfactory selectivity, yielding poor extraction efficiency. More also, DESs components comprising of choline chloride (ChCl) as HBA in conjunction with citric acid and oxalic acid as HBD have proved to be suitable solvents for the extraction and recovery of bioactive compounds such as phenolic acids from different plant sample matrices such as Morus alba L., and Aegle marmelos respectively. Interestingly, the organic acid-based DESs showed the highest efficiency probably because of their polarity, making them a suitable choice for extracting and recovering polar molecules like phenolic acids and anthocyanins, as summarized in Table 2.
Researchers have also investigated, [36] evaluated and applied five DESs, including choline chloride ChCl as HBA, while malic acid, glycerol, sorbose, and oxalic acid were selected as HBD for the recovery and extraction of phenolic analytic from the grape skin waste. The extraction method yielded good extraction efficiency, with Anthocyanins being the most major phenolic derivatives present in grape skin extract. Also, the mixture of ChCl-oxalic acid DES (1:1) and 25% water showed excellent extraction efficiency towards the phenolic compounds, followed by the other two organic acid-based DESs. Subsequently, these same researchers investigated and evaluated an optimized DES-based UAE approach for extracting and recovering anthocyanins in wineless. However, this time, only three DES, namely: sugar, polyalcohol, and organic acid-based DESs, were employed. The optimized UAE method based on ChCl-malic acid DES showed excellent extraction efficiency, so it was selected as the best extraction solvent. However, the Organic acid-based DESs showed better recoveries for anthocyanins than the sugar polyalcohol-based DESs [37].
The flavonoids
Flavonoids are low molecular weight antioxidants involved in diverse biological and physiological processes that aid defensive mechanisms against abiotic stress by conferring different protective pigmentation and patterns to fruits and flowers of plants. They exhibit a common molecular mechanism that inhibits angiogenesis and cell proliferation, modulates transcription and its kinases, and downregulates endogenous antioxidants [38]. Flavonoids consist of two benzene rings in their chemical structure connecting via a pyrane ring, such as flavane; others include chalcones flavanones, flavones, isoflavones, proanthocyanidins, flavonols, flavanol, and anthocyanins as shown in Figure 3a-g. They are capable of arrays of remarkable biological activities like anti-atherosclerotic, antimicrobial, and antimicrobial activity against Escherichia coli and Salmonella.
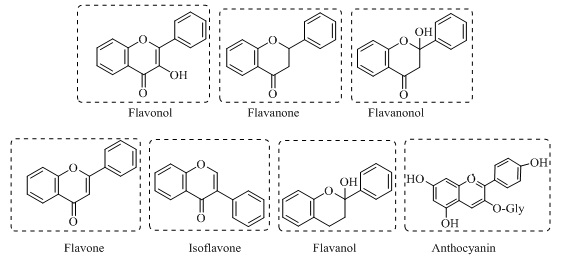
Figure 3. Chemical structure of different flavonoids
Tannins
These are natural products of plant origin that possess many phenolic rings in their chemical structure. They are classified into condensed and hydrolyzable tannins, as demonstrated in Figure 4. These condensed tannins contain flavonoids molecules with varying degrees of condensation, while the Hydrolyzable tannins contain a mixture of phenols linked with ester in their chemical structure originating from hydrolysis by mineral acids, alkaline compounds, and enzymes.
 Figure 4. Chemical structure of tannin
Figure 4. Chemical structure of tannin
Anthocyanidins
These consist of an aromatic ring linked to a heterocyclic ring through a carbon bond [39]. They exist naturally in the form of glycoside, thus making this kind of phenolic compound complex. They enrich and increase antioxidant activities in fruits. They are powerful antioxidants that neutralize oxidant molecules produced from cellular embolism pathways and free radicals. They are of tremendous benefits and protection to the health of cells, tissues, and the whole plants.
Terpenes
This type of aromatic compound is grouped owing to its characteristic carbon skeleton. They are characterized by fragrances from many fruits, leaves, seeds, wood, flowers, and roots, especially in herbal and fruit-flavored wines. They are a diverse significant class of natural or plant-derived organic hydrocarbons present as a major constituent of resins (turpentine) in conifers or evergreen plants. They are simple volatile organic compounds consisting of carbon and hydrogen only, but most importantly, they possess unique antifungal, antibacterial, anticancer, and antiviral properties, making them essential ingredients in pharmaceuticals, perfumes, food, and cosmetic industries. They exist in plants as defensive mechanisms giving offensive odors to discourage herbivores and insects from attacking or appealing to their predators.
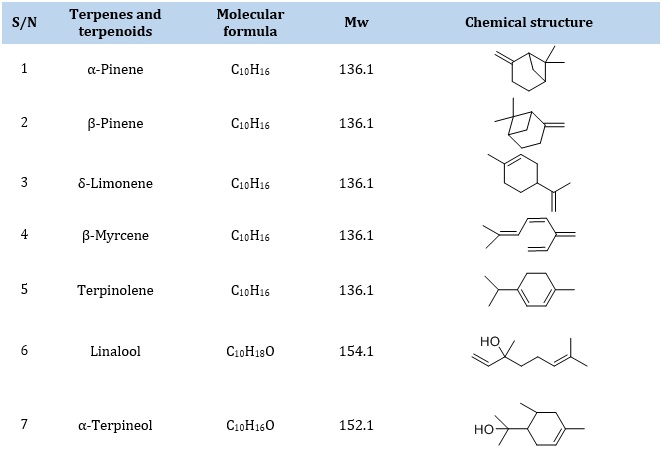
Terpenes and their derivatives are important constituents in essential oils derived from plants, trees, and flowers, which consist of basic five-carbon isoprene units as seen in Table 1, for example, 2-methyl-1,3-butadiene. Terpenes are organic hydrocarbon compounds comprising more than one isoprene unit (a five-carbon block) called hemiterpenes (a single isoprene unit), monoterpenes (two isoprene units), sesquiterpenes (three isoprene units), diterpenes (four isoprene units), sesterterpenes (five isoprene units), triterpenes (six isoprene units), sesquiterpenes (seven isoprene units), tetraterpenes (eight isoprene units), and polyterpenes (many isoprene units). These isoprene units are linked together with another isoprene in head-to-tail chain reactions [40].
Terpenoids are polar, non-volatile, or semi-volatile secondary metabolites derived from multicyclic groups of terpenes and oxygen. Moreover, terpenes and terpenoid precursors are dimethylallyl diphosphate and isopentenyl diphosphate, and they are ingredients for drugs, fragrances, and flavors. They are mainly isolated from their natural matrices using non-polar ScCO2 solvent [41].
Table 1. Selected Terpenes and terpenoids bioactive compound

Synthesis of bioactive compounds
Secondary metabolites or bioactive compounds can be synthesized in four major pathways, namely, Mevalonic acid (produces terpenes), non-mevalonate (MEP) (produces terpenes), Shikimic acid (produces aromatic amino acids and Phenolic compounds), and malonic acid (produces Phenolic compounds only) pathways as shown in Figure 5.
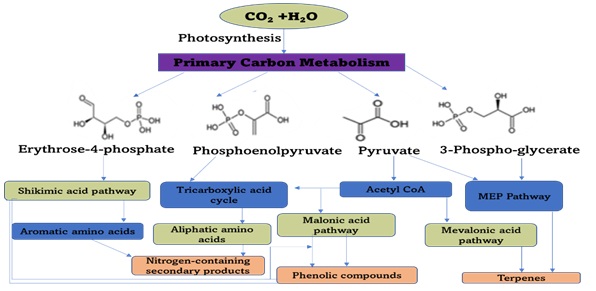 Figure 5. Schematized synthesis pathways for plant bioactive compounds
Figure 5. Schematized synthesis pathways for plant bioactive compounds
It is better to build a standard approach to integrate the varieties of plant species and their bioactive compounds, especially those that carry significant health benefits and therapeutic advantages. Although some integrated approaches have been reported in the past, it is necessary to show the sequence of these medicinal plants starting from the sample preparation to their clinical trials and eventually to large-scale commercialization. As shown in Fig 6 illustrates the work plan to study medicinal plants with the suitable techniques needed for separation, isolation, extraction detection, structural and elemental characterization, and diverse suitable conditions that favor excellent and effective extraction and good extract yield. The nature and impurities associated with different bioactive compounds in their natural environment make extraction and isolation complex. This also requires meeting various conditions and extraction approaches that enhance their selective recovery from their natural source. The objectives of all these techniques are to (a) extract targeted analytes (bioactive compounds) from complex environmental matrices (medicinal plant), and (b) enhance the effective sensitivity and the selectivity of the individual analytical approach used. (c) increased concentration of the targeted analytes extracted by preparing a highly sensitive bioassay (d) transform and purify the extracted bioactive compounds into a more suitable or derivatized form to ease separation and detection, finally, (e) provide eco-friendly, quick, reliable, reproducible, and rugged method regardless of varying concentrations in the sample matrices.
Bioaccessibility and biocompatibility of bioactive compounds
The health benefits of bioactive compounds depend primarily on their availability and the amount consumed. Primary sources of these bioactive compounds are seafood, cereals, meat, plants, fruits, and vegetables. Phytosterols such as campesterol, sitosterol, and stigmasterol are present in plant cells. When consumed, they help to prevent the effect of low-density lipoprotein cholesterol in the human body. Also, Glucosinolates are bioactive compounds available in cruciferous plants, which are an integral part of the Mediterranean diet. They are characterized by specific tastes and odors, such as broccoli, cabbage, cauliflower, and Brussels sprouts [42]. They possess compounds like Sulforaphane, responsible for healthy functions, such as chemo-preventive, anti-cancerous, and anti-inflammatory effects. Other bioactive compounds are the fructooligosaccharides and Dietary fiber inulin, which contribute immensely towards the microbiota symbiosis homeostasis in human guts, aids lipid metabolism, and infant nutrition, regulates blood sugar level, and lowers obesity and colon cancer [42]. The Polyunsaturated fatty acids, which include eicosapentaenoic acid, docosahexaenoic acid, and linoleic acid, are bioactive compounds present in food, oils seeds of soybean, walnuts, sunflower, flax, corn, and fish (mackerel. herring, and salmon). These prevent the risk of different diseases such as functional gut environment, muscle wasting disorder, blood pressure homeostasis, and metabolic syndrome [42].
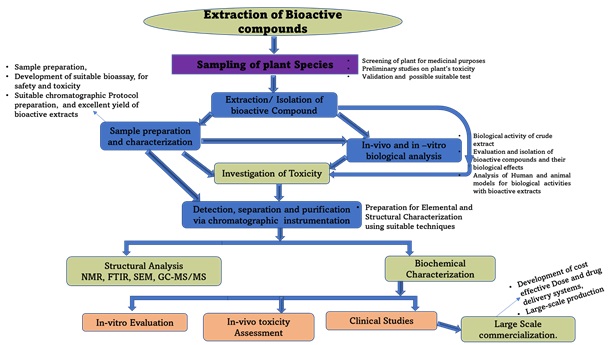 Figure 6. Schematized workable plan for the extraction of plant bioactive compounds from sampling industrialization.
Figure 6. Schematized workable plan for the extraction of plant bioactive compounds from sampling industrialization.
Bioaccesibility
This is the available number of bioactive compounds that contributes towards food metabolization. These bioactive compounds interact with food constituents such as lipids, carbohydrates, and proteins to alter the rate of metabolism [42].
Bioavailability
This is the ability of these bioactive compounds to be broken down and distributed throughout the human body. Parameters that aid the metabolism and distribution of bioactive compounds include solubility, the complexity of structure, degree of polymerization, mass transfer, and molecular interactions [43]. According to the reported literature, four main factors affect the bioavailability of bioactive compounds. These include the bioaccessibility and stability of the bioactive compound at the injection time, the fractions that remain biologically active after passing through the gastrointestinal digestion to the absorption site, and the remaining fraction of the bioaccessible compoundsre absorbed into the epithelium cells [43].
NADES-Related Extraction of Bioactive compounds
The use of DES, either the HDES or NADES, for extraction of various analytes, especially macromolecules, is made possible because of its valuable dissolution capabilities and other tunable properties to enhance its extraction prowess [44]. These NADES are commonly supported by heating and many modern extraction approaches to assist the extraction of different phytochemical bioactive and active organic compounds like terpenes, phenols, and the like from environmental samples, making it greener and faster [45]. Bioactive moieties like terpenoids, carotenoids, phenolic compounds, and fatty acids are majorly synthesized from natural sources, especially plants, and are used in pharmaceuticals, food, cosmetics, and other fields of study. The largest of these groups are the carotenoids responsible for the orange, green, and yellow coloration/pigmentation in flowers, fruits, and vegetables. Lycopene and β-Carotene are subsidiaries of carotenoids and are essential food ingredients because they produce different colors [41]. They are easily extracted from their natural sources using scCO2, the most conventional solvent. Others, like tocotrienols and tocopherols, are associated with anticancer and antioxidant properties, thus making them essential ingredients in the food and cosmetic industries. These groups of phytochemicals are generally isolated from vegetable oils [46].
In the last decade, there has been growing interest in the use of DES and HDES for extraction and separation processes, especially in the recovery and extraction of bioactive compounds, thus indicating the excellent potential can extraction capability in the extraction of bioactive compounds from a food source [33]. Different types of DES systems, such as NADES, HDES, and binary and tertiary systems, are suitable and can be used to extractioactive molecules based on their physicochemical properties and the properties, source, and nature of the targeted analytes [33]. Also, optimum conditions of other parameters, such as extraction temperature and time required to enhance the extraction efficiency, should be investigated on different DESs under the same conditions before evaluating the most suitable choice.
There have been different reports on the effect of different solvents, such as ethyl alcohol, n-hexane, and methanol, on the extraction of bioactive compounds from various parts of the plants (seeds and leaves). Effective extraction of pure concentration of phenolic compounds from the plant requires different solvents with different polarities. However, highly polar solvents such as methanol are sensitive and effective in extracting pure and high concentrations of antioxidants. Researchers have also reported the use, effectiveness, and suitability of N, N dimethylformamide (DMF), acetone, and methanol for excellent yield of antioxidants, especially, phenolic compounds from walnut fruits. According to the report, N, N dimethylformamide (DMF), acetone, and methanol, these solvents' prowess and extraction capacitytood out compared to ethanol [47]. In another report, as shown in Tables 2 and 3, Methanol, water, acetone, and ethanol were used to extract the bioactive compounds from Ivorian plants. It was observed that ethanol gave a better extract yield compared to others. Many solvents have also been solely used to extract of phytochemicals and bioactive substances from the dried powdered plant, thus eliminating the interferences caused by water [48,49]. Also, adding water to the DES by more than 50% can cause a dilution effect by reducing its viscosity and altering polarity, invariably leading to lower extraction yields [50]. Extraction solvents have been applied to extract different bioactive compounds such as alkaloids, terpenoids, phenolic compounds, and polysaccharides [51]. These phenolic functionalities include lignan, flavonoids, phenolic alcohol, and phenolic acids. In recent times, Kanberoglu et al reported the use of mixtures of HDES tetrabuthylammonium bromide/decanoic acid and tetrabuthylammonium chloride/decanoic acid for the extraction of quercetin from grape, tomato, apple, and onion [52]. Similarly, researchers have proposed using designer solvents involving 1:2 ratios of selected HDES combinations involving choline chloride/butyric acid, choline chloride/caprylic acid, and choline chloride/valeric acid for the extraction of piperine from peppercorns and ground pepper samples (black pepper). Also, researchers have prepared different HDESs using a 1:2 ratio combination of decanol, dodecanol, butanol, octanol, methyltrioctylammonium chloride, tetrabutylammonium bromide as HBDs and choline chloride salts as HBAs for the extraction of terpenes from clove, cumin, nutmeg, cinnamon, thyme, and fennel [53,54].
Antioxidant-related properties of NADES extracts
The capability of NADES to successfully extract and isolate valuable products and derivatives without altering the antioxidant capacities is novel. Several assays are used to evaluate the quantity of the antioxidant properties of natural product extracts. These include total antioxidant capacity (T-AOC), superoxide radical scavenging activity, 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay, ferric reducing antioxidant power (FRAP), and hydroxyl radical scavenging activity [55].
In an experiment reported by Pavic and co-workers [56] on the extraction of antioxidants from rue Ruta graveolens L. using NADES (ChCl-CA (2:1). The antioxidant activity was reported to be< 80% (72.53%). In contrast, the same group reported the use of ChCl-ascorbic acid (2:1) on the extract from Mangifera pajang fruit waste, where <12g (AEAC)/g) of ascorbic acid was evaluated [57]. In another experiment, seven NADES mixtures were synthesized as follows: ChCl-oxalic acid dihydrate (1:1), ChCl-tartaric acid (1:1), ChCl-glycolic acid (1:3), ChCl-LA (1:1), ChCl-malic acid (1:1), ChCl-CA monohydrate (1:1), and ChCl-malonic acid (1:1) were utilized in the extraction of Spruce (Picea abies) prior to evaluation of their antioxidant potential. It was reported that a remarkable antioxidant potential of 16.6%, 15.49%, 13.18%, 16.59%, 12.16%, 11.34%, and 15.42% was evaluated. These incredible feats attained by NADES extracts bring their potential to the limelight, making them research hotspots and replacing the use and application of conventional organic solvents in various fields. The increase in the yield of NADES extract considerably increases the antioxidant potential of the extracted or targeted compound. Thus, water content, the molar ratio, and the types of NADES used alters the antioxidant activity and extract yields [57,58]. In another report, the extraction of antioxidants from flowers, leaves, and rhizomes of Curcuma longa L using different ratios (1:2, 2:1, 1:1) of menthol or chlorine chloride and lactic, lauric acid and acetic acid has also been investigated [59].
Antibacterial-Related Properties of NADES Extracts
The highly beneficial potential of NADES extracts to inhibit the growth of bacteria and fungi has made the development of novel, eco-friendly, and designer solvents possible and beneficial to many industries. The antibacterial activity of all NADES-extracted bioactive compounds is investigated and evaluated using the disk diffusion approach against various bacterial strains (gram-positive and gram-negative). In an experiment reported by researchers on the investigations of the antibacterial activities of NADES extracts from rue leaf against Bacillus subtilis, Pseudomonas aeruginosa, Escherichia coli, and Staphylococcus aureus [60]. All NADES extracts from the plant showed good antibacterial potential against the tested bacteria strains. In contrast, the extract displayed 125 µg/ml minimum inhibitory concentrations against all the strains except against P. aeruginosa, which was evaluated to show the best antibacterial efficiency with a unique MIC value of 62.5 µg/ml.
NADES affect the biological activities of the compounds extracted, extract yield, quantity, and characteristics of the bioactive content. Hence suitable NADES specific to the corresponding target analyte must be developed to produce an excellent molecular interaction.
Conventional and non-conventional extraction Techniques for Bioactive compounds
Conventional method
The extraction efficiency of any conventional method is based on the choice of solvents and the polarity of the targeted compounds. These factors should be evaluated based on cost, application of co-solvent, eco-friendliness, the molecular affinity between the solvent and solute, mass transfer, human and environmental safety, and toxicity. Bioactive compounds are plant materials extracted by different solvents concerning their extraction potency and application of heat [32]. The suitable solvents adopted in this approach are water, HDES, and NADES like ethanol, acetone, methanol, petroleum ether, n-hexane, and others. The basic techniques employed to obtain plant bioactive extracts include Hydro-distillation, percolation, decoction, Soxhlet extraction, infusion, and maceration. This conventional approach is low capital intensive, easy to perform, and the technology readiness level (TRL) ratio is 9:10. However, their demerits include high energy, toxic solvent, and time consumption, resulting in adverse occupational, environmental, and health hazards. Significantly, the risk of deterioration of thermal sensitive constituents from vegetable and fruit sources are probable.
Soxhlet extraction
German researcher Franz Ritter Von designed the process and apparatus (Soxhlet extractor)o extract valuable lipids and bioactive compounds from different natural Environmental media [85]. It is also used as a medium to compare the extraction capacity, purification, and concentration of the active bioactive compounds to that obtained from the new and modified process. This is a repeated process involving a thimble, distillation flask, siphon, and extraction solvent. A known amount of dry samples was placed in the thimble, and both were placed in the distillation flask containing the suitable extraction solvent. The siphon aspirates the solvent and unloads it back into the distillation flask. This repeated process begins and ends the extraction procedure. The solvents extract the bioactive compounds from the sample and elute them in the distillation flask. The solution carries extracted solutes from the solid bed plant and dumps it into a distillation flask. After extraction equilibrium, the bulk liquid containing the solvent and extracts is evaporated to dryness. Consequently, many experimental methodologies involve using ES to isolateseful bioactive moieties from diverse natural matrices [86].
Maceration
This is the low-cost, small-scale, traditional approach to extracting essential oils and bioactive compounds from plants. The process of Maceration takes place in a closed vessel, beginning with grinding the dried plants to enhance the surface area and ease of mass transfer from the sample to the menstruum (solvent). After that, the filtration of the occluded liquid after pressing the marc to elute the solvent and the Eluate, thus removing all impurities. There is a need for repeated shaking to enhance the extraction process by increasing the rate of diffusion and to allow the addition of new solvent to the marc to further elute the concentrated eluate from the marc while simultaneously removing the concentrated solution from the sample surface [87]. The shortcomings of this approach are the lower extraction efficiency and time-consuming extraction process [88,89].
Notwithstanding, it can be used to isolate heat-labile compounds, thus suppressing the development of probable microbes before fermentation. Also, it alters the successive synthesis of flavor producing-yeast during fermentation. Researchers have reported using the maceration technique to successfully extracthenolic derivatives such as anthraquinone from Rheum emodi [90].
Hydro-Distillation
This physicochemical process involves the extraction of essential oils and bioactive compounds without employing organic solvents. Generally, this complete one-step extraction approach involves dehydration of the plant sample matrices by either water distillation, direct distillation, or water and steam distillation. The major physicochemical processes in this approach comprise hydro-diffusion, hydrolysis, and decomposition by heat [3]. The process of hydro-distillation begins with the packaging of the sample matrices into a still compartment (tea bag), after that, an adequate amount of water at 100 oC is required. Alternatively, steam can be injected into the system (directly in the sample) in place of the boiling water to influence the extraction of the bioactive compounds by leaching it from the plant tissue. There is a need to cool the system indirectly. Hence the cold water is pumped into the system over the condenser to cool the mixture of water vapor and oil. Thus, the condensed mixture gravitates towards the automatic separator, where the bioactive compounds and essential oil are automatically separated from the water. The demerit of this process is the heat required, and this can result in the loss of volatile and thermos-labile components. However, it is fast and does not utilize organic solvents [91].
Liquid-liquid extraction
This approach is renowned for solvent extraction and is suitable for azeotropic mixtures and temperature-sensitive compounds. This extraction approach involves the mass transfer and molecular integration between the sample and two immiscible liquid phases, namely: the aqueous and the organic phase. The extraction success depends on the dissolution of the target analytes (bioactive compounds) in the organic phase. The plant sample matrices are mixed vigorously and shaken with the immiscible liquids in a separating funnel. After some time, two distinct liquid layers are formed in which the analyte of interest (bioactive compounds) distributes itself between the aqueous and organic phases based on the relative solubility of the immiscible solvents [92]. This approach does not require distillation. However, it has some demerits, the formation of emulsion, requiring a large amount of organic solvent, and highly laborious and difficult automation [93].
Infusion
This method is suitable for extracting soluble and volatile bioactive compounds from pomace, leaves etc. The samples are macerated and soaked in aqueous or organic solvent (cold, warm, and hot) for a few hours. The liquid extracts containing the bioactive compound are after that concentrated via a rotary vacuum evaporator at low temperatures. Generally, this process is faster than maceration. However, it varies depending on the solvent used, also the nature and type of waste [94,95]. Researchers have also reported the use of this process for the isolation of about 36% of valuable bioactive compounds from medicinal plants [96]
Decoction
This is the quickest procedure employed when preparing kwath or quath known as ayurvedic extracts, which are later concentrated, filtered, and processed to be consumed orally. This method is suitable for extracting thermal or water-soluble bioactive compounds from seeds, stems, and other hard wastes. The solid waste was initially pulverized into powder, after that, the powdered material is boiled for close to an hour in a closed system using a suitable quantity of water until the water in the mixture evaporated. Subsequently, the boiled sample mixture is filtered to remove the solid residue [95]. The targeted bioactive moieties are primarily soluble in oil. Researchers have also discovered higher efficiency in extracting phenolic and antioxidant compounds by combiningnfusion and decoction of Teucrium polium L. from the Lamiaceae spp [97]. This simple process is characterized by some demerits such as longer extraction time, requires large amounts of solvent, and doesn’t have palpable flavor. Others include difficulty in storage and transportation [98].
Percolation
This is a continuous process of extracting bioactive compounds by repeatedly passing fresh solvent through the samples until a saturated solvent containing bioactive moieties is collected. This process is more efficient than the maceration approach because the approach is repeated. Originally, the dried sample matrix is conditioned using a suitable solvent for less than 4h prior to complete immersion in the solvent for at least 24h. At the end of the repetitive extraction cycle, the percolator is then repeatedly filled with fresh solvent (water or organic) until maximum recovery of the extracted bioactive compounds is obtained. Generally, the demerits of this approach include the consumption of large amounts of solvent and extraction time [95].
Others include mild extraction approaches like serial exhaustive extraction (SEE) and conventional reflux extraction. The former involves fractionating non-polar extracts with solvents of increasing polarity to enhance the optimum extraction of bioactive compounds. The demerit of this process is the degradation of heat-labile bioactive compounds due to excessive heating [95]. While the latter is unsuitable for extracting heat-labile natural bioactive compounds, it’s more effective and efficient when compared with maceration and percolation techniques. The merits of this approach, as shown in Table 4 include saving of extraction time andrequiring less solvent. Researchers have also reported using this method to extract total phenolic compounds from dried fruit (Terminalia chebula Retz). At optimized extraction conditions, this reflux approach also shows a greater yield of baicalin and puerarin from the plant [99].
Non-Conventional Green Extraction Approach
The demerits of the conventional approach, as revealed in Table 4 as high cost, high power consumption, longer extraction time, evaporation of a large amount of solvent, thermal decomposition of heat-labile targeted constituents requiring highly purified solvent, and lower extraction selectivity. To overcome these demerits, there is a need to develop and introduce pristine, eco-friendly, and promising non-conventional extraction approaches such as pressurized liquid extraction, microwave-assisted extraction [100], pulsed electric field assisted extraction, hydrodynamic cavitation-assisted extraction [101], ultrasound-assisted extraction [102], supercritical fluid extraction [46], enzyme-assisted extraction [103] and microextraction approach. These have also been identified and reported as excellent energy sources to enhance extraction efficiency, reducing overall separation time, and enhancing product quality [104]. In addition, the conglomeration of extraction approaches using two or more greenness approaches offers more excellent separation of bioactive [105]. Others are instantly controlled pressure drops [102], instantly controlled pressure drops [106], and cloud point extraction [107]. These eco-friendly approaches comply with the use of safe solvents auxiliaries, renewable feedstock, and designer safer chemicals, involving less hazardous chemical synthesis that are economical, consume less time and energy, and reduce derivatives, waste, and impurities, thus, preventing atom economy, degradation, pollution, and accident.
Ultrasound-Assisted Extraction (UAE)
This procedure requires the use of compressed and expanded sound waves beyond human hearing (20 kHz to 100 MHz) to collapse and grow bubbles in cavitation[108]. This process of cavitation produces a large amount of energy by converting the kinetic energy of motion into heat energy required to heat (5000 K) the bubble content [3]. The principle of cavitation is experienced only by liquid and liquid-containing solid composite. Thus, facilitating the mass transfer and solvent access between organic and inorganic compounds elution from the plant matrices. The UAE process involves two types of physical extraction mechanisms: diffusion and scattering across the cell wall of the plant materials and elution by raising the cell material's bioactive content after breakinghe cell walls [102]. The decomposition of tissue bubbles closely follows this rupture and cell wall damage resulting from the implosion of expansion and then micro-shooting, which results in erosion, surface peeling, and particle disintegration [108]
The effectiveness and efficiency of the UAE principle are governed by the temperature, pressure, moisture content of the sample, particle size, time of sonication, and milling degree. Also the UAE can be employed in conjunction with other classical extraction approaches, as shown in Tables 1 and 2. Several advantages of this method include a reduction in the use of solvents, energy consumption, and time. This is because the UE facilitates mass transfer, energy transfer, consolidating mixing and selective extraction, reduced extraction time and temperature, reduced equipment size, and thermal gradients, thus making extraction fast and less laborious[109-112]. Researchers have reported using UAE to effectively extract four bioactive isoflavone derivatives from herbal plants. These isoflavone derivatives include genistin, daidzin, malonyl, and glycinin, while four isoflavones were isolated from soy plants using a similar approach, improving better sensitivity, accuracy, and excellent extraction efficiency [113]. The extraction was combined with a mix-stirring approach with different extraction times and solvents. In another experiment, Phenolic compounds were extracted from a strawberry using a semi-automatic method based on UAE between 0.8 s duty cycle for 30 s. The phenolic functionalities include ellagic acid, naringenin, kaempferol, rutin, and naringin. In another experiment, UAE was employed to extract and recover chlorogenic acid from fresh, dried barks, and fresh leaves using optimized parameters: 70% methanol at solvent: sample ratio (20:1) within 30 min [114,115].
In another experiment, optimized parameters of UAE were employed to extract phenolic compounds and anthocyanins from the grape peel. The optimized extraction parameters were based on extraction time, solvent, and temperature [116]. The optimized extraction of Rosmarin acid, Phenolcarboxylic acid, and carnosic acids from Rosmarinus officinalis based on the Ionic- liquid-based UAE approach has also been reported to be characterized by high extraction efficiency and shorter extraction time when compared with conventional methods. Another developed method for the optimized UAE extraction of triterpene saponins (ginsenosides) from the genus Panax plant (ginseng), based on the use of ternary DES mixture consisting of 30% water and sucrose, glycerol, and l-proline (1:9:4). The SPE procedure was performed using a cartridge of Oasis®HLB (hydrophilic-lipophilic (50:50) sorbent). Later, 6 mL of ethanol was utilized to recover the ginsenoside prior to characterization by the HPLC-UV, while the DES was rinsed and washed from the column with 6mL of water. The resultant mixture of water and DES was freeze-dried to recover and recycle the DES. This extraction was done thrice with a minimum of 82% satisfactory recovery efficiency. The DES-based UAE method has become a hotspot in research in recent times because after evaluation and optimization of the extraction parameters, such as selection of suitable DES for the targeted analytes, irradiation time, extraction temperature and time, amount of water added, the efficiency of extraction and greater extract yield. Although water reduces the viscosity of the DES, the increase in temperature generally increases the extractive yield. However, increased temperature and ultrasonic time can degrade the targeted thermal-labile analytes [33,35,36][117-119].
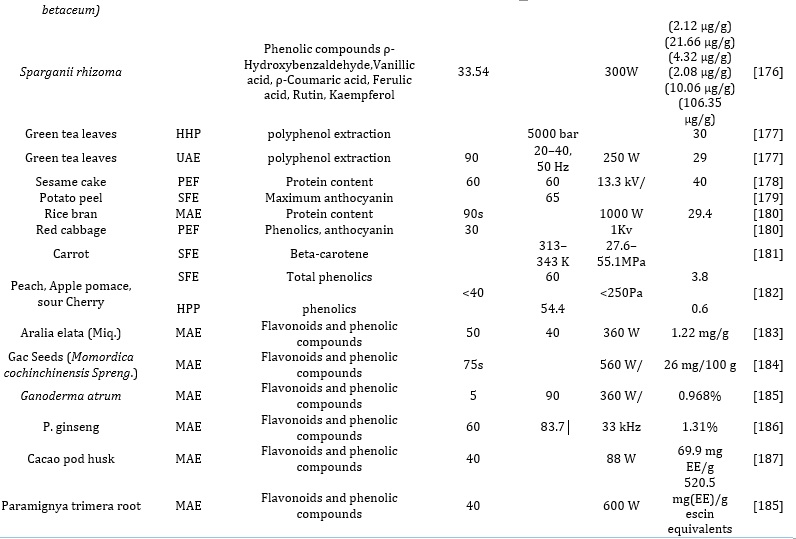
Pulsed-Electric Field Extraction (PEF)
The PEF method was developed based on the dipole nature of the membrane molecules and the charge in the cell membrane separated by an electric potential subjected to it. Thus, improving the drying, pressing, diffusion, and extraction process. Unlike the UAE, which breaks the cell walls of the plant material to ease extraction, the PEF destroys the cell membrane in the suspension via an electric field potential. The introduction of the increased transmembrane potential (1 V) causes repulsion between the charged molecules, resulting in the formation of pores on the cell membrane's weak areas, invariably resulting in increased deleterious permeability. The process involves using exponential decay pulses in conjunction with a simple circuit that includes two electrodes in a chamber operating in a batch or continuous mode to treat the plant materials [120-122]. Summarily, the PEF increases the mass transfer during the extraction process by first destroying the plant membrane structure to release the intracellular compounds from the plant tissue and aiding the cell membrane permeability, thus enhancing the extraction efficiency and decreasing the extraction time. The effectiveness and the efficiency of the PEF approach are altered by the strength of the applied electric field, inherent properties of the plant materials, pulse number, treatment temperature, and the specific energy input. The demerit of this approach is the destruction of the cell membrane of the plant tissue even at low temperatures and moderate electric field range 0.5-1kV/cm within 10−4–10−2 s. However, this reduces the damage and degradation of the thermal-labile bioactive compounds. Hence it is preferably used as a pretreatment approach in conjunction with another conventional extraction method to lower the extraction effort. It has also been reported by researchers that in the solid-liquid extraction of bioactive compounds such as betanin from beetroots by applying the PEF pretreatments techniques (1kV/cm with low energy consumption of 7x103Vkg-1) and when compared with mechanical pressing and freezing, the approach shows a remarkable degree of extraction efficiency and greater extract yield. Also, in another experiment using the same method for the recovery of phytosterols from maize and daidzein and genistein was extracted from soya beans. It was observed that the extraction efficiency increased from 32.4% and 21% for both maize and soya beans, respectively. In another experiment, researchers reported using the PEF pretreatment method for the extraction of anthocyanin mono glucosides derivatives from grapefruits, as shown in Tables 1 and 2. The method applied yielded good extraction efficiency for the recovery of anthocyanin derivatives from the grape. Also, it reduces the extraction time, making the reaction less laborious by eliminating the maceration of the skin step. Also, this process enhanced the stability of the bioactive derivatives, such as polyphenols and anthocyanin, from the vinification step. Also, the pretreatment of merlot skin via PEF pretreatment enhances the extraction and recovery of anthocyanins and polyphenols. Thus, making these permeabilization techniques outstanding. The Pasteurization of varieties of food items like milk, dairy products, juice, liquid eggs, and soup has been extensively demonstrated using this approach [122]. Researchers have also reported extracting phenolic compounds from borage, sesame seed cake, and spearmints at optimum experimental conditions, such as 99 pulses of 3 kV/cm at 4102 ± 239 J/kg energy input using the PEF, is better compared with treatments involving microwave and heat. Elevated energy inputs increase the protein, polyphenol contents, and disintegration index. Although, this is not recommended for solid meals [122].
Enzyme-assisted extraction (EAE)
In routine extractions, solvent inaccessibility to penetrate unaided when dispersed into the cell cytoplasm of plant matrices to extract and recover phytochemicals or some retained polysaccharide-lignin compounds is a result of the presence of hydrogen or hydrophobic interactions [123]. This necessitated the development of a novel and effective enzymatic pre-treatment approach to dissociate and release the bounded compounds, thus increasing the extraction efficiencies and extract yield. The inclusion of α-amylase, pectinase, and cellulase enzymes hydrolyses the structural lipids bodies and polysaccharides, enhancing the breaking of cell walls during extraction. This approach enhances recovery and extract yields [124]. The enzyme-assisted extraction technique is categorized namely: enzyme-assisted cold pressing and enzyme-assisted aqueous extraction. The later technique (EAAE) was developed mainly for the routine extraction of oils from various seeds. While in the former (EACP) technique, the hydrolysis of the seed cell wall is enhanced by enzymes in the absence of polysaccharide-protein colloid in the system. However, the polysaccharide-protein colloid is present in the EAAE technique. Also, the EACP technique is characterized by non-inflammable and non-toxic properties. In the EAE technique, the extraction efficiency is determined by the particle size of the plant materials, the concentration and chemical composition of the Enzyme applied, hydrolysis time, and solid-to-water ratio (moisture content). However, the entire plant cell wall hydrolysis is practically impossible for EAE with the prevalent enzymatic preparation techniques, also making the entire process expensive. Moreover, this approach has significant technical and commercial setbacks, such as the high cost of large-scale production, especially when it involves elevated enzyme-to-to-substrate ratio. In addition, the procedure also limits the yield in the extraction of Phyto-bioactive compounds [125,126].
Researchers have reported the use of the EAE method for the extraction of phenolic phytochemicals from grape pomace fruits during the production of wine. It was also discovered that the degree of plant cell wall breakdown by enzymatic activities is proportional to the total phenolic composition yield. The extraction and recovery of total phenolic contents from mandarin, orange, Meyer lemon, grapefruit, and Yen ben lemon via the EAAE approach using cellulolytic enzyme MX amongst other enzymes, yielded a good yield, especially with the use of cellulolytic enzyme MX. Other researchers have extracted non-anthocyanin flavonoids, phenolic acids, and anthocyanins from grape pomace using a mixture of the cellulolytic and pectinolytic enzymes (2:1) and reported an enhanced overall increase for all the bioactive compounds extracted compared to sulfite-assisted approach. In another recent experiment, researchers also reported using enzymatic hydro-alcoholic and non-enzymatic extraction approaches to extract phenolic compounds from raspberry solid waste. The enzymatic hydro-alcoholic approach yielded good extraction efficiency and extract yield. In another experiment, the use of xylanase, cellulase ,celluclast, novoferm, hemicellulase, and pectinex enzymes in the EAE approach for the extraction of phenolic antioxidants from grape waste and bay leaves yielded a good extraction efficiency, especially with the strong effect of novofern on the extraction yield [127]. The mixture of amyloglucosidase and α-amylase enzymes enhanced the yield of curcumin from turmeric [128].
The Solid Phase Extraction (SPE)
This pretreatment approach is widely adopted for the isolation, purification, and extraction of the targeted analyte. Solid-phase extraction (SPE) was launched in 1975 and became commercially valuable in 1978 [129,130], and since then, it has been widely used for the extraction and pretreatment of environmental samples [131]. The SPE uses solid materials with highly selective adsorption capabilities to extract the target analytes or eliminate complex interfering components from the environmental sample matrixes. SPE can be divided into the Normal-Phase (here the sample solution is non-polar while the solid stationary phase is polar), Reversed-phase (stationary phase is hydrophobic), Mixed-Mode or Ion-Exchange (mode of separation is by partition coefficient interaction between the sorbent (stationary phase) and analytes), MIP (molecularly imprinted polymer (MIP)) and other Special techniques (cartridges-doped with reagent) [131]. In the SPE, functionalized absorbents are utilized in the LC Column, and these include inorganic sorbents such as magnesium and alumina silicate, silica gel, charcoal, polymers, and nanocomposites, including CNTs (carbon nanotubes), novel graphene, and more recently, MIPs (molecularly imprinted polymers). The carbon nanotubes can be grouped into single-walled and multi-walled CNTs characterized by inherent features like excellent mass transfer property, large surface area, ease of design and modification, and effective and efficient adsorption capability. The absorption of this carbon-based sorbent to organic compounds depends on mainly non-covalent forces like hydrogen interactions, electrostatic forces, van der Waals, and hydrophobic interactions. The SPE technique is governed by the partition coefficient between the sorbent and the target analytes in the sample solution. The sorbent is closely packed inside microcolumns, cartridges, or syringe barrels. The stepwise procedure of the extraction includes (1) column conditioning, (2) sample loading, (3) washing, and (4) elution. The disadvantages of the arrangement of the SPE column's design include cartridge channeling, clogging of the used cartridge, and leaching of the sorbent, invariably leading to a prolonged extraction process and time.
Nan and co-researchers reported investigating and evaluating a solventless extraction and recovery of flavonoids from the DES extract of Flos sophorae using the SPE method. Also, the recovery of rutin from the same plant using reversed-Phase cartridge-based SPE was evaluated. Extract from the Flos sophorae was loaded inside the SPE column, and methanol and water were utilized to elute the retained rutin and polar DES component mixture, as shown in Table 2 [132]. The eluate was there after being filtered, hydrolyzed, and characterized using a suitable chromatographic technique such as the HPLC-UV. This method yielded excellent percentage recoveries (92%) for the rutin from DES. Compared with the anti-solvent extraction method concerning kaempferol, quercetin, and isorhamnetin glycosides, the percentage recoveries ranged from 81-87%. Researchers have also reported using other microextraction techniques, such as liquid phase microextraction and solid phase microextraction, with prominent merits such as consumption of a low volume of solvent, eco-friendly, and requiring a minimal amount of sample[133]. As reported by Aydin and colleagues, after the successful development of a liquid-liquid microextraction approach for the extraction of curcumin from an herbal tea sample using a Choline chloride-based DES in conjunction with UV–visible spectrophotometer. At optimum conditions, the recoveries ranged from 94% to00% [134]. The Solid Phase micro-extraction approach combines sampling, extraction, and enrichment procedures into one single step, thus reducing consumption in time and solvent [135]. Other researchers have also reported the use of the micro-solid phase extraction method (μ-SPE) in conjunction with NADESs/DESs/HDESs for the extraction of bioactive compounds in complex environmental matrices, especially from plants and food samples, as shown in Table 2
The Microwave Assisted Extraction (MAE)
This extraction mechanism is the most frequently employed novel approach for extracting different bioactive molecules and other soluble products from diverse raw materials into a fluid via microwave energy from electromagnetic fields in a range of 300 MHz to 300 GHz frequency [64]. This system comprises two oscillating fields (magnetic and electric) that are perpendicular to each other. The direct impact of the waves (electromagnetic energy) on polar materials is converted into heat energy after dipole rotation and ionic conduction mechanisms. During the ion conduction mechanism, the frequency of change in ion directions along the fields is orchestrated by the resistance to the medium flow. This increases the collision between the molecules and invariably generates heat. Summarily, the heat generated in the MAE involves three consecutive processes, namely: Separation of the solutes from available active sites on the complex sample matrices altered by increased pressure and temperature [136,137]; More also, the diffusion of extraction solvent across sample matrices and lastly, the recovery and release of solutes (targeted analytes) from the sample matrix to the extraction solvent. Researchers have also reported the optimized extraction of tannin, cinnamaldehyde, E, and Z-guggulsterone from various plant matrices. Also, using the same approach, bounded phenolic acids were released from sorghum, maize bran, and flour. Also, researchers have reported the optimized extraction of rich polyphenolic compounds from onion peel waste using ChCl:Urea: H2O (NADES), assisted with Microwave power (100–300 W). The extraction efficiency of the valorization process was based on microwave power, time, and liquidto-solid ratio. The Optimized MAE approach was rapid and easier, yielding better extraction efficiency than other conventional methods. For optimization of the extraction method and maximization of the flavonoids and phenolics acid recoveries, the following parameters were optimized: extraction time, solvent concentration, and microwave power via design experts to enhance the extract's electron donating ability. This green method of extraction (MAE) is characterized by notable merits over other conventional methods, such as enhanced extract yield, consume fewer organic solvents, reduced thermal gradients, reduced equipment size, and faster, better extraction of bioactive compounds from plant sample matrices enabled by heat [68]. However, it is selective to the extraction of organometallic and organic compounds. The extraordinary ability of NADES and HDES to exert numerous connections with bioactive compounds such as π−π, electrostatic interactions, and hydrogen bonding have exceeded the actions of conventional solvents [68]. Also, they are highly efficient and effective in initially disrupting the organized structures of natural matrice to enhance the mass transfer and solubility of the targeted bioactive compounds [138]. For example, as summarized in Table 1 and 2, researchers have reported the extraction of phenolic bioactive molecules from mulberry leaves via an MAE-assisted approach by NADES that are methanol-based, (ChCl/glycerol (1:2)) and ethanol-based [139]. Other applications are summarized in Table 3.
Table 2. Applications of NADES and HDES as an extraction media for bioactives
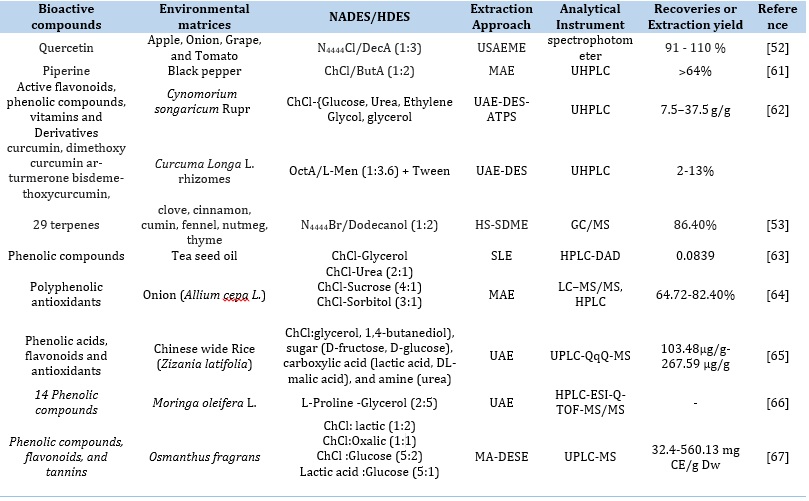


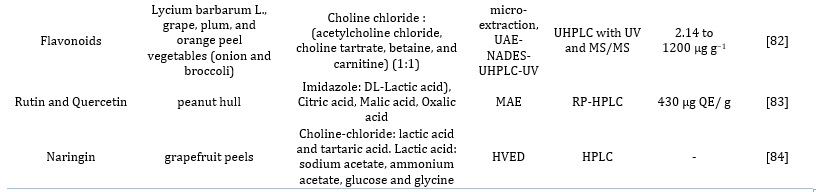
Table 3. Optimized conditions for selected sample matrices for the extraction of bioactive compounds
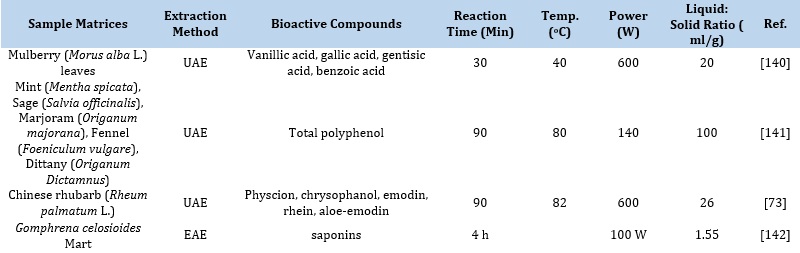

Pressurized Liquid Extraction (PLE)
The Pressurized liquid extraction (PLE) approach was first introduced by Richter and colleagues in 1996 with different names such as accelerated fluid extraction (ASE), high-pressure solvent extraction (HSPE), and enhanced solvent extraction (ESE) [188]. The Automated PFE approach utilized high pressure and high temperature to enhance rapid extraction by promoting the solubility of the targeted analyte, thus, aiding the mass transfer rate with increased solubility. Also, the increase in pressure and temperature reduces the amount of solvent consumed, decreases surface tension and viscosity and use of the solvent, and reduces extraction time but invariable increase the extraction rate [189]. This is better when compared to the Soxhlet extraction because it saves time and reduces solvent usage and consumption. Also, PFE is a better alternative to supercritical fluid extraction because of its ability to effectively extract polar compounds and organic pollutants from complex environmental matrices, especially those stable at high temperatures. Also, the extraction of bioactive compounds from marine sponges and the extraction and recovery of natural products from complex environmental matrices, consumes little quantity of organic solvent. Hence the approach is profoundly accepted and tagged green [189]. This approach is unsuitable for heat-labile phenolic compounds because elevated temperatures will affect their functions and structure [190].
Optimized conditions of green extraction of bioactive compounds such as isoflavones from various plant materials, especially freeze-dried soybeans, without any form of degradation, have been reported in the literature. Also, there have been reports of the extraction of sterols and terpenoids from tobacco with Soxhlet extraction compared with ASE and UAE based on reproducibility, solvent consumption, extract yield, and extraction time. It was discovered that the PLE is considered better with a rapid extraction process and lower solvent consumption. Researchers have also reported the PLE extraction of flavonoids from spinach utilizing a mixture of ethanol and water (70:30) as solvents at temperatures ranging from 50–150 C. Optimized PLE extraction of Amaryllidaceae alkaloids such as galanthamine and lycorine from Narcissus jonquilla was reported to be more effective compared to UAE and MAE. Also, using the same method, phenolic compounds such as epicatechin, caffeic acid gallocatechin (GCT), myricetin, catechin, chlorogenic acid, and epicatechin gallate were extracted and recovered from the bark, stem, leaves, seeds, and flowers of Anatolia propolis at 40°C, 1500 psi for 15 min [191,192]. Also, as shown in Table 3, researchers have recently reported and compared the use of MAE with conventional solvent extraction of bioactive polyphenols from dried sea buckthorns involving NADES and HDES.
Supercritical Fluid Extraction
This approach was first introduced for extraction by Hannay and Hogarth in 1897 but made public and pronounced by a renowned scientist Zosel for decaffeinating coffee, for which he was credited with patency in 1964 [193]. This approach consists of a mobile phase CO2 tank, pressure-sized gas pump, oven that houses the extraction chamber, pump and co-solvent, and control valve with flowmeter attached to alter or maintain high pressure in the trapping vessel entire system. This garnered much attention over the years from researchers in various fields of study such as nutraceutical, polymer, pharmaceutical, and food industries, especially in the decaffeination of coffee. The supercritical state is significant because substances, regardless of the state of matter, are subjected to pressure (Pc) and temperature (Tc) beyond their critical points and form supercritical fluids, where their specific properties and state vanish and cannot be recovered with modifications in pressure or temperature. These supercritical fluids are characterized by gas-like properties such as surface tension, solvation power, diffusion, liquid-like density, and solvation power, making it an all-important fluid suitable for extracting bioactive compounds within a short time with excellent extraction efficiency and greater yield. The merits of this approach include:
- Repeating the extraction via refluxing the SFE to the sample is possible and adequate to complete the extraction.
- The SFE is characterized by higher diffusion coefficients, lower surface tension, and viscosity than any liquid solvent, thus making mass transfer favorable and enhancing deep penetration into the sample matrices.
- The selectivity of the SFE is impacted by altering the temperature (Tc) and pressure (Pc), thus making the solvation potential of SFE greater than that of any liquid.
- The SFE is notably a quick, eco-friendly approach because it consumes little solvent, requires a small amount of sample, and depressurization saves time.
- The SFE approach experimented on at room temperature is ideal, suitable, and compatible with extracting heat-labile compounds.
- SPE approach provides adequate information about the mechanism and extraction process, which can be tunable to optimize the extraction; also online- coupling with other analytical chromatographic techniques makes the approach preferable and suitable for highly volatile compounds.
- SF's use, reuse, and recycling in the SFE approach reduce the wastage and cost of producing new SF.
Researchers have also reported the limitations of this approach, as seen in Table 4, such as that it is costly, consumes high power, requires elevated pressure for operations, and no polar substance can be extracted. In another report, SFE-CO2 was employed for the optimized extraction of phyto-bioactive moieties from plant parts (leaves of radish plant), yielding excellent extractions of flavonoids, total phenolic and antioxidants at optimum pressure and temperature of 400 bar and 40 C respectively [194]. Other researchers have reported the extraction of coumarins, diterpenoids, flavonoids, phenolic compounds, and other beneficial constituents from plants like T. chebula, rosemary, Catharanthus roseus leaves, Bulgarian yarrow, Wedelia calendulacea, Feijoa leaves and sage. At optimum conditions ranging from 40 -50 C, pressure ranged 250-6000 pounds per square inch (psi), and dynamic extraction time ranged 60-90 min, there were increased yields of the beneficial bioactive compounds, including oxygenated monoterpenes, indole alkaloids, terpene alcohols camphor, sesquiterpenes, borneol and artemisia ketone [195].
Back Extraction Method
This approach involves applying a DES biphasic solvent system to extract target analytes from complex environmental systems. The choice of solvent is critical to the success of this approach because the solvent used in this approach must be immiscible to the aqueous extract solution and have an affinity to the analyte of interest. Xu and colleagues introduced this approach to recover target analytes (flavonoids) from DES extracts on citrus peel waste [196]. Ternary DES used in this experiment was formed from the mixture of 20% water and ChCl–levulinic acid–N-methyl urea (1:1.2:0.8) (v/v) in conjunction with UHPLU-MS/MS for the extraction of the methanol extract. The approach yielded an excellent recovery for molecules with different polarities such as kaempferol-3-O-rutinoside, hesperidin, and narirutin flavonoids (GoFs) and polymethoxylated flavonoids (PMFs) such as nobiletin, 5-hydroxy-6,7,8,3′,4′-pentamethoxyflavone, tangeretin, sinensetin and 3,5,6,7,8,3′,4′-pentamethoxyflavone. Other solvents such as chloroform, ethyl acetate, n-butanol, n-hexane, and acetone were also utilized in a 2-step back extraction to evaluate their extraction and recovery efficiencies on selected GoFs and PMFs such as hesperidin and tangeretin and nobiletin respectively. This two-step backward extraction involves forming a biphasic system by adding ethyl acetate to the DES solution of flavonoids mixture prior to vigorous shaking and centrifugation. The percentage extraction and recovery efficiencies were 95% and 87% for PMFs (nobiletin, tageretin, and hesperidin) in the organic phase (n-butanol) and DES solution (water and DES)[196], respectively. The method was also reported by Tan and colleagues for the recovery of flavonoids from the extract of citrus peel only than Ionic liquid was used in place of DES [197].
In another report, Hang and colleagues also reported applying the two-step back extraction approach for the recovery of flavonoids (apigenin and luteolin) from celery. A 10 mL of ethyl acetate was added to the DES extract, and the mixture was vigorously mixed and shaken for 15 min at RT to enhance the separation of the analytes. The method yielded a good percentage recovery for both apigenin and luteolin at 109 and 83%, respectively [198].
Table 4. Advantages and differences in the non-conventional methods for the extraction of bioactive compounds
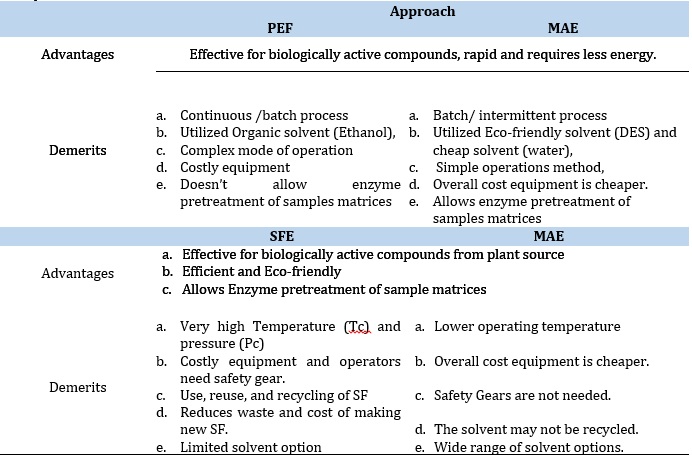

Other methods are briefly summarized below, such as high hydrostatic pressure (HHP) assisted extraction, subcritical-water extraction, and high voltage electric discharge. The HHP is one of the recently developed approaches that is receiving more research interest and concentration owing to its beneficial properties in the pharmaceutical and food preservation industries. Summarily, this non-invasive process involves forcing (200- 600 MPa) fluid through a very tight gap value resulting in cavitation, turbulence, and high shear stress that distort the matrices microstructure, thus resulting in a homogeneous, smaller-sized particle of the extracted bioactive compounds with enhanced physical, structural and textural properties. This approach's merit is that it requires no heat. Hence thermal degradation of the bioactive components does not occur. Also, the inherent covalent bonds are not damaged. However, it may lead to cell deformation, damage to the cell membrane, and incomplete denaturation of protein, leading to structurally fragile materials [199]. Researchers have also reported the optimum extraction of bioactive compounds (p-hydrobenzaldehyde, vanillin, and ferulic acid) from Bifidobacterium longum and Bifidobacterium longum [200,201]. The SWE (superheated water) is a greenness approach that requires the use of hot liquid water or pressured water to extract bioactive compounds. The merits of this approach is cost-effective and efficient for extracting non-polar compounds. At optimized conditions such as temperature range of 100-370 °C and Pressure 10–60 bar) and with the presence of NADES varieties of polyphenols, polar and non-polar moieties like hesperidin, pectins, and narirutin have been isolated from T. chebula fruit apple, citrus peels, and pomace [202-205]. Also, the extraction of the following bioactive compounds has also been reported from plants like thyme, Verbena, and rosemary, such as microalgae (Chlorella vulgaris), algae from Cystoseira abies-marina, Undaria pinnatifida, Porphyra spp., Sargassum muticum, and Sargassum vulgare. This process also includes therm-oxidation, caramelization, and Maillard. Finally, the HVED approach is a green, novel, and efficient approach that can be classified into three processes, namely: batch, continuous, and circulating systems, which involves the three basic processes such as high electric field creation, cell death, and augmentative mass transfer [206]. In this process, two electrodes (needle-plane, point-plane, or point-plate) are submerged into the extraction media containing the plant material and the NADES, creating and releasing energy directly into it to produce a plasma channel with destroyed cell tissues, which allows more beneficial chemical compound and derivatives to be isolated from the plant and by-products. They type of electrode used is based on the operations of this process. Hence, the structural variation in the devices alters the concentrations of the electric fields [207]. The merit of this approach includes shorter treatment time, does not allow the use of toxic solvents, does not require heat. However, it is a non-selective extraction approach [208]. Researchers have used this approach to extract phenolic compounds and polyphenols from spent coffee and grape stems under optimized conditions of 0–40 kV and 0–50 Hz. The system comprises a treatment chamber, a high-voltage pulse generator, an extraction tank, and a pump conveyance unit [209].
Physiochemical Properties of NADES
The selection of suitable chemical constituents and the nature of NADES is necessary to design a defined system and target analytes. The ability to tune their properties and potential to specified requirements make these solvent novel and open to various promising possibilities and applications in various fields like nutraceutical, food, pharmaceutical, and cosmetic industries [210]. The physicochemical parameters critical to extraction techniques include solubility, viscosity, pH, and polarity. Altering these parameters immediately affects their linear relationship and extraction efficiencies. For example, dilution or reduction in viscosity of the DES can decrease diffusivity, thus, improving the extraction efficiency [211-213]. Similarly, researchers have also reported the effect of using NADES and HDES with different viscosities on the extraction efficiency of anthocyanins from plants like Catharanthus roseus. The report also confirmed that extraction efficiency and viscosity are inversely correlated [214] and that the physicochemical properties such as density, viscosity, conductivity, and polarity are also influenced by the presence of waterClick or tap here to enter text. [215,216].
Viscosity
The viscosity of NADES is a function of the individual constituent that makes it up, and dilution by the addition of water can significantly reduce or impact it. Also, altering the water content can alter the conductivity of the NADES. The application of temperature can also impact the positive linear relationship between viscosity and temperature [213]. The HDESs exhibit a wider range of viscosity values (7–86, 800 mPa*s), which hypothetically allows for the alteration or modification of the extraction process. However, the non-ionic HDESs show lower viscosity, approximately less than (<219 mPa*s)[217,218]. Also, no defined rule allows the prediction of the viscosity of HDESs, even when combining HBA with different molar ratios of HBDs. Researchers have reported that viscosity can depend on the nature of HBA given the following example: Quaternary Ammonium Salts (QASs)> menthol > thymol > long chain fatty acid [219]. In addition, increased temperature affects the viscosity of the DEs, causing disintegration of the inherent electrostatic links between the DES moieties. Thus, elevated working temperatures are inversely proportional to viscosity.
Polarity
The polarity of the individual solvent component in NADES affects the propensity to extract or recover polar or non-polar compounds. The addition of water to NADES can impact its performance in the extraction of polar compounds. For example, if the water content is high, the NADES performs better for extracting polar compounds, but when the water content is low, the extraction of weak or low polar compounds is enhanced. When considering the extraction of hydrophilic and polar bioactive compounds from natural environmental matrices, water is considered a suitable and effective extraction and recovery solvent, although this can be energy-sapping. However, the percentage of extraction efficiency with organic solvent can also increase but has some demerits that limit its application, such as toxicity, being not eco-friendly, being expensive, and low biodegradability [220,221].
Interestingly, the concentrations of NADES components in human daily diet intake are increasing daily. Also, the like of acetone, methanol, ethyl acetate, water, and ethanol, have demonstrated a classic extraction efficiency prowess for elemental species, low polar, and polar compounds over traditional solventsClick or tap here to enter text.[51,222]. In addition, the compatibility and suitability of the NADES to renowned analytical separation, spectrometric and instrumentation techniques such as TLC [223], UV–Vis , HPLC [224][225][33], DAD [33], ICP-OES, and mass spectrometry [225] [33] is evident in the satisfactory results reported. These NADES have also been evaluated and validated for immunoaffinity, and their addition to electrolytes has also contributed immensely to the electrochemical detection of natural products such as antioxidants and phenolic compounds [. This smart extraction strategy involving NADES has been exploited and adopted in the new and recently developed microextraction technology such as the µ-SPE, and DES-LPME because it is cheap, safe, and efficient. For example, the formation and use of micro and nano-sized DES dropletsmicro and nano-sized DES droplets in aES-based-LPME extraction and preconcentration of rhodamine B before further characterization and determination via micro-cuvette UV-VIS spectrophotometer [119]. In another experiment reported by Wang and co-workers on the use of NADES in conjunction with static-headspace gas chromatography (SHS-GC) for detecting residual solvents in drugs and drug-related substances [226]. The NADES-SHS-GC approach was reported to be characterized by high sensitivity. The SPME is a widely used microextraction technique based on solid sorbent. This approach also combines the preconcentration and extraction process into one step, where the sample matrices is functionalized with NADES, and the stationary phase is attached to the fiber dispersed into the solvent matrices medium to which the partition equilibrium is measured. Like the SPME extraction of phenolic compounds and alkaloids from Larrea Divaricata plant leaves via the SPME -MW-NADES approach [227]. The modification and functionalization of nanomaterials is a physicochemical process that involves adding new functional groups, and this is another promising possibility credited to DES. The newly modified DES nanomaterials are characterized by STA, FTIR, XRD, TEM, Raman spectroscopy, and SEM and can be utilized in diverse health-related, environmental applications such as biosensors, wastewater treatment, and drug delivery [228].
pH
This critical parameter must be considered and carefully evaluated when synthesizing NADES. Generally, the degree of acidity of a NADES eutectic mixture depends largely on the presence of acidic components, its nature, and the type of HBD. pH, Chemical potential, and molecular interactions in DES are altered by the availability of cations and anions and hydrogen bonding between the ions with other constituents in the solvents. In any experimental procedure, the pH of the targeted analyte must be higher or relatively close to the pKa values, because analytes are easily isolated from the environmental matrices in a neutral form using weak polar solvents. On the contrary, isoelectric points are considered for extracting target analytes like protein because they exhibitmphoteric properties [229]. Also, highly polar compounds require organic acid-DESs, NADES, and acidic environments for optimum extraction.
In an experiment reported by various researchers while evaluating the pH of different NADES eutectic mixtures such as Bet-MA (1:1), ChCl-Gly (1:2), ChCl-proline-MA (1:1:1) and ChCl-MA (1:1), it was observed the addition of water to the organic-based NADES mixture and the availability of acidic constituents in the HBD contributed to the pH increase except for NADES Bet-MA (1:1) and ChCl-proline-MA (1:1:1) eutectic mixtures.
Cytotoxicity
From the evaluation and investigation of the NADES, the molar ratio and composition of its inherent constituent contribute mainly to the toxicity of NADES [44]. Although the precursors to NADES are from natural compounds, their Eutectic mixtures are non-toxic. However, this should be ascertained and investigated for their toxicity so as not to affect the application of the formed NADES. Hence, the reason why cytotoxicity is adequately investigated and evaluated towards different cells of human beings. The effect, properties, contributions, and cytotoxicity of NADES have been evaluated towards human breast cancer (MCF-7) cells [44, 51], human cervical cancer cell line (HelaS3) [44,51], catfish ovary (CCO) fish cell [113], human ovarian cancer (CaOV3) cell, and animal cells such as mouse fibroblast-like (L929) cells [128] and mouse skin cancer (B16F10) cell [113]. This experiment has helped to develop new green NADES that are safe, suitable, and eco-friendly. The use of Choline chloride (ChCl), a distinguished HBA, has been discovered to be toxic to the cells of human embryonic kidneys [215] (HEK-293). Also, the formed eutectic mixtures can be more toxic that the individual precursor [225]. This can result from the emergence of seas of delocalized charges during the formation of the hydrogen bonding between HBA and HBD while synthesizing the NADES. Also, chemicals with localized charges are less toxic compared to delocalized ones.
Conclusion
Conclusively, the advent and deleterious increase of pandemic infections and diseases has necessitated the build-up of body immunity to combat them and the green synthesis of bioactive compounds from natural environmental matrices (plants). This has tremendously increased the interest in research and investigations on using NADES for sustainable, eco-friendly, and rapid extraction and recovery methods for the bioactive compounds from the plant. The significant and novel properties of these NADES have significantly impacted this extraction and many other applications and extraction techniques across all fields. However, these extraction and recovery methods are not without challenges because of numerous experimental conditions which can be optimized and inherent properties of the NADES (viscosity, pH, molar ratio, polarity, and density) and bioactive compounds which are tunable. Also, the demerits and merits of extraction techniques have increased the combination of some of the effective conventional extraction methods, thus, eliminating some of the demerits, enhancing the extraction, and increasing yields towards maximum bioactive compounds without noticeable degradation. Finally, the development and conjunction of new methods should be developed, evaluated, optimized, and implemented based on the inherent properties of the plant matrices, choice of bioactive compounds towards efficient, eco-friendly, sustainable, fast, large-scale, sensitive, and sophisticated extraction and recovery techniques for bioactive compounds and derivatives in years to come.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgment
This work was financially supported by the Tertiary Education Trust, TETFUND Nigeria, and The Federal Polytechnic Offa, Kwara State, Nigeria. The authors deeply appreciate this support.
Disclosure statement
The authors declare that they have no conflict of interest
Orcid
Aduloju E. Ibukun : 0009-0009-5468-6283


