Document Type : Original Research Article
Authors
Department of Applied Chemistry, Ardabil Branch, Islamic Azad University, Ardabil, Iran
Abstract
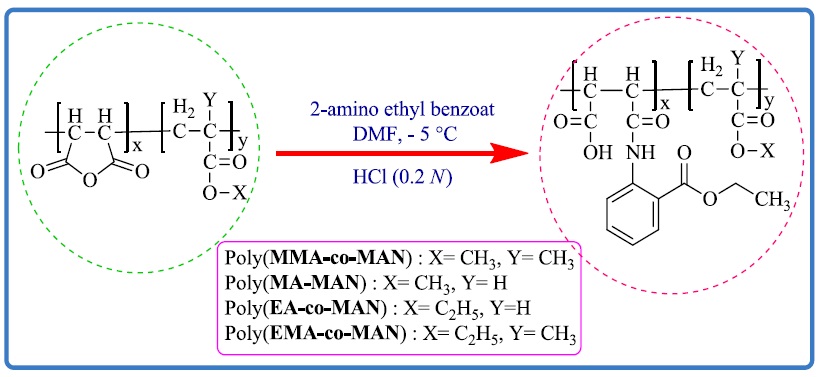
Maleic anhydride (MAN) copolymers with methyl methacrylate, ethyl methacrylate, methyl acrylate, ethyl acrylate, and butyl acrylate monomers at 1:3 mole ratios were synthesized by free radical polymerization in the presence of azobis(isobutyronitrile) as initiator and dry ethyl acetate as solvent. Copolymer compositions were obtained using related proton nuclear magnetic resonance (1HNMR) spectra, and the polydispersity of the copolymers was determined by gel permeation chromatography. Then, a solution of 2-amino ethyl benzoate salt as a nucleophilic reagent reacted through the ring opening of anhydride groups in copolymers resulting to modified copolymers Iab to Vab. All the prepared polymers were characterized by Fourier transform infrared and1H NMR spectroscopic techniques. The glass transition temperature (Tg) of all copolymers before and after modification was determined by dynamic mechanical thermal analysis (DMTA). It was shown that chemical modification of MAN copolymers with 2-amino ethyl benzoate substituents as side chains decreased the free volume of the polymers, and therefore, the rigidity and Tg are increased.
Graphical Abstract
Keywords
- Chemical modification
- Infrared spectra
- dynamic mechanical thermal analysis (DMTA)
- thermo-gravimetric analysis (TGA)
- Maleic anhydride (MAN) copolymers
Main Subjects