Document Type : Original Research Article
Authors
- Mahmood Payehghadr 1
- Samaneh Adineh Salarvand 1
- Farzaneh Nourifard 1
- M. Kazem Rofouei 2
- Niloufar Bahramipanah 1
1 Department of Chemistry, Payame Noor University, 19395-4697 Tehran, Iran.
2 Faculty of Chemistry, Kharazmi University, Tehran, Iran, Iran.
Abstract
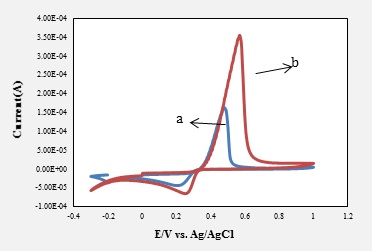
A new ligand, 1,5-bis( para methoxyphenyl)-3- ethyl-1,4-pantaazadiene has been synthesized by reaction of the 4-methoxyaniline (p-anisidine) with ethylamine. The mixture was stirred in an ice bath for 30 min. The structure of the synthesized compound resulted from the IR and 1HNMR and 13CNMR. Afterwards, a carbon paste electrode modified with this new ligand was developed for the silver determination at nanomolar level concentration. The electerochemical properties of this modified electrode was investigated by employing cyclic voltammetry (CV) and differential pulse voltammetry (DPV) methods in an acetate buffer solution (pH=4.8). The effect of pH, scan rate, percentage of modifier and buffer as supporting electrolyte on the electrode process were investigated. The oxidation peak of Ag+ was observed at about 0.4- 0.5 V. The resulting electrode demonstrated linear response across a 10-9 to 10-8 mol.L-1 of silver concentration range with a detection limit value of 1.61×10-10mol.L-1, on the basis of a signal to noise ratio of 3. Relative Standard deviation (RSD%) of the electrode performance was 2.41%. This MCPE, were showed high sensitivity and selectivity to Ag+ ions in aqueous samples.
Graphical Abstract
Keywords
Main Subjects