Document Type : Original Research Article
Author
Chemical and electrochemical treatment, Mineral Processing Dept. Central Metallurgical Research and Development Institute, CMRDI P.O.Box 87 Helwan 11421 Cairo, Egypt
Abstract
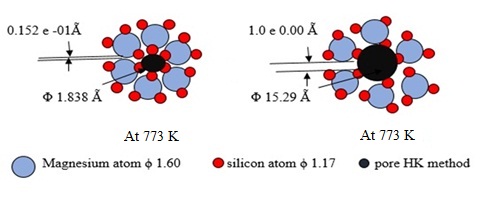
This study reports on the thermal dislocation and crystal growth changes during preparation of active metasilicate of group 2 periods 3.4 and 5 elements from waste fines of finishing process of crystal glass (CG) industry. Sodium carbonate fused the fines at ≈1273 K for 120-180 minutes followed by water leaching. Alkali silicate goes into solution. Primary chloride and sulphate salts of elements of group 2 periods 2, 4 and 5 added to the alkali silicate solution at temperatures 300 K to 353 K for 60 minutes to give metasilicate as a white precipitate followed by heating at 473 K to 1073 K for 30 minutes. The study discussed the physical changes taking place after heating. Results revealed that surface area and adsorption capacity of Mn ions from underground water increased with the surface area of the silicate associating the decrease in atomic radius of the element. The effect of pH, concentration of the reactants and temperature on the synthesis and physical changes of the silicate salts has been investigated. Formation of the silicate salt proceeded in a reaction sequence involving atom rearrangement to form a honeycomb structure with very narrow pore diameter amounting to 1.538 Ằ. Heating silicate at 773 K dislocate the oxygen atoms around metal atoms to form unit crystals with mild pore diameter of 15.29 Ằ. With further increase in temperature, narrow pore system collapses to form sponge structure with wider pore diameter and lower surface area. The manuscript tabulated the physical changes in digital presentation derived mathematically.
Graphical Abstract
Keywords
Main Subjects