Document Type : Original Research Article
Authors
1 Department of Pharmacy, Gyan Ganga Institute of Technology and Sciences, Jabalpur, (MP) 482003, India
2 Shri Ram Institute of Technology, Pharmacy, Near ITI Madhotal, Jabalpur, (MP) 482002, India
3 Medicinal Chemistry Research Laboratory, SLT Institute of Pharmaceutical Sciences, Guru Ghasidas University, Bilaspur, (CG) 495009, India
Abstract
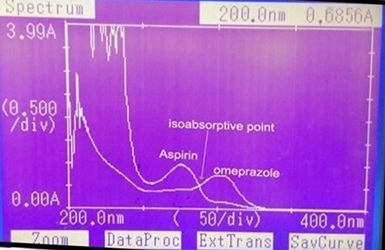
The present study deals with the development and validation of two simple, accurate and precise spectrophotometricmethods for estimation of Aspirin (AS) and Omeprazole (OMP) in their binary mixture. The first method was developed using Vierordt’s simultaneous equation method. It involves absorbance measurement at 257 and 300 nm (λmax of AS and OMP) in methanol. Second method involves the formation of Q-absorbance equation using the respective absorptivity values at 284.5 nm (isoabsorptive point) and 300 nm (λmax of OMP). The drugs obey Beer’s Lambert’s law in the concentration range of 4-20 µg/mL and 2-10 µg/mL for AS and OMP respectively for both the methods. Limit of quantitation for Vierordt’s method for both the drugs was found to be 0.85 µg/mL, whereas for absorbance ratio method it was 0.68 and 0.88 µg/mL for AS and OMP respectively. % recovery for both the drugs was in the range of 99.78-100.31% indicating excellent accuracy for both the methods. The methods were precise, with a relative standard deviation of less than 1.25% for both drugs. The developed methods were validated according to ICH guidelines and values of accuracy, precision and other statistical analysis were found to be in good accordance with the prescribed values. Thus, both methods can be used for routine monitoring of drugs in industry for the assay of bulk drugs and commercial formulation (Yosprala).
Graphical Abstract
Keywords