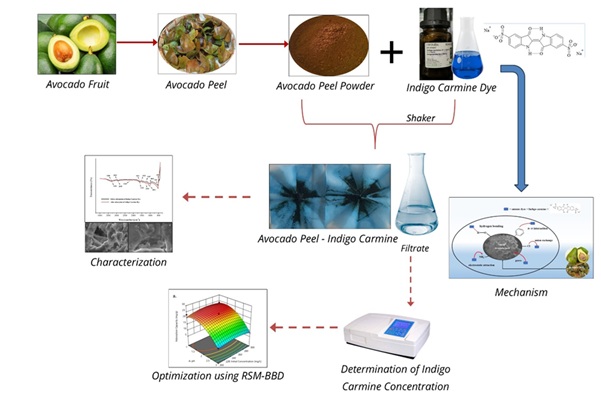
Eco-Friendly Removal of Indigo Carmine from Aqueous Solutions Using Avocado Peel: A Response Surface Methodology Approach
Pages 556-576
https://doi.org/10.48309/ajca.2026.549396.1940
Corry Handayani, Adewirli Putra, Emriadi Emriadi, Refilda Refilda, Rahmiana Zein, Joshua O. Ighalo
Abstract Avocado peel (Persea americana Mill.), an abundant organic solid waste, was utilized as an eco-friendly biosorbent for the removal of Indigo Carmine (IC) dye from aqueous solutions. The adsorption process was optimized using Response Surface Methodology (RSM) based on a Box–Behnken Design (BBD), considering four key variables: solution pH, initial dye concentration, contact time, and temperature. The optimal conditions were determined to be pH 1.46, dye concentration 263.4 mg/L, contact time 95.48 min, and temperature 55.01 °C, yielding a maximum adsorption capacity of 25.17 mg/g. Characterization using FTIR, SEM–EDX, and BET analyses confirmed the involvement of surface functional groups, significant changes in surface morphology, and pore-mediated adsorption. The adsorption behavior followed the Freundlich isotherm model, indicating multilayer adsorption on a heterogeneous surface, and the pseudo-second-order kinetic model, suggesting that the uptake rate depends on both active sites and adsorbate concentration. Thermodynamic parameters indicated that the process was spontaneous and exothermic. The adsorbent demonstrated excellent cost-effectiveness for water treatment applications, achieving a cost performance of 0.539 USD/g-dye. These findings highlight avocado peel as a sustainable, low-cost, and environmentally benign biosorbent for industrial dye wastewater treatment.