M. Vijayarathinam; A. Kannan; P. Akilan; V. Chanrasekaran; T. Gunasekaran
Abstract
A succinct total synthesis of an indoloquinoline alkaloid was achieved through a one-pot, two-step process involving a cascade of sodium hydride and aniline, followed by a base-mediated ...
Read More
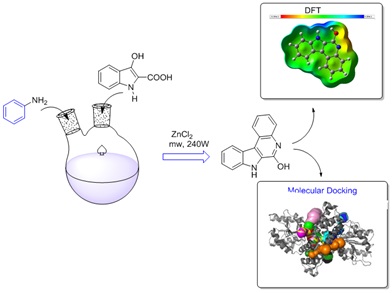
A succinct total synthesis of an indoloquinoline alkaloid was achieved through a one-pot, two-step process involving a cascade of sodium hydride and aniline, followed by a base-mediated Claisen condensation reaction. Subsequently, the reaction was conducted via microwave-assisted synthesis (MAS), resulting in a good yield of the title compound with eco-friendly methodologies. The structural properties of the synthesized compound were validated using FT-IR, 1H-NMR, 13C-NMR, and GC-Mass spectral analyses. In addition, FT-IR approaches were employed to compare the vibrational wavenumbers obtained through simulation with experimental wave numbers. Furthermore, DFT calculations were employed to fine-tune the structural parameters and investigate FMOs, Mulliken atomic charges, MEP, and NLO properties, and molecular docking studies evaluated its N3 binding site of the primary coronavirus protein 6LU7. Likewise, the title compound is assessed for its ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) properties, revealing promising pharmacokinetic profiles devoid of any detected indications of non-toxicity.