Dina Naseer Ali; Ahmed Ahmed; Alaa Hussein J. Alqaisi
Abstract
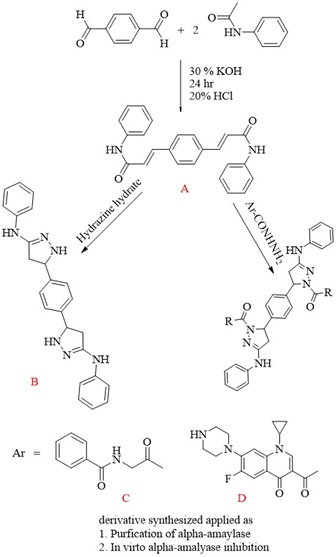
This study focuses on synthesizing three pyrazole derivatives, namely B, C, and D. Fourier transform infrared spectroscopy (FTIR) and proton nuclear magnetic resonance spectroscopy ...
Read More
This study focuses on synthesizing three pyrazole derivatives, namely B, C, and D. Fourier transform infrared spectroscopy (FTIR) and proton nuclear magnetic resonance spectroscopy (1H-NMR) were used to study the synthesized derivatives. Pyrazole derivative B was produced from the chalcone derivative (A) reaction with (4-nitrophenyl) hydrazine. The pyrazole derivatives C and D were produced from the reaction of chalcone derivative (A) with hydrazide derivatives (N-(2-oxopropyl)benzamide and 3-acetyl-1-cyclopropyl-6-fluoro-7-(piperazine-1-yl)quinolin-4(1H)-one), respectively. The Purification of α-amylase was conducted on pancreatic cancer patients from Iraq, employing three distinct purifying techniques. The experiment yielded a high level of enzyme activity, specifically 7 U/mL and a specific activity of 8.75 U/mg protein. These results were achieved using an ammonium sulfate saturation ratio of 65%. The graph shows a peak in enzyme activity at 3.75 U/mL in the elution area. This peak, fraction 55, has 11.3 U/mg protein activity. The stability of α-amylase was constant over a pH range of 5.0 to 9.0. In the pH range of 6 to 7, enzyme stability is highest at pH 7. At pH 5 and 9, strength decreased. The enzyme was less active at pH 5 and 6 but more active at pH 7 and 9. However, enzyme activity peaked at pH 8.0. The α-amylase enzyme maintained 100% activity at 27-37 °C, demonstrating stability. However, enzyme activity decreased to 50% at 47 °C when temperature increased. The inhibitory effect of bis-chalcone and pyrazole derivatives increased with concentration. The results show that compound B has the most significant inhibitory efficacy, at 50%.