Document Type : Original Research Article
Authors
Department of Chemistry, Faculty of Physical Sciences, Bayero University Kano, Kano, Nigeria
Abstract
One cannot overstate the importance of looking for alternative solutions and effective corrosion control in our ecosystems. To investigate the corrosion inhibition behavior of ethanol extract of Prosopis juliflora leaves on mild steel in various concentrations of tetraoxosulphate VI acid (H2SO4) solution, electrochemical technique, scanning electron microscopy (SEM), and Fourier transform infrared spectroscopy (FTIR) were used in this research. The weight loss data showed that the highest inhibition effectiveness was seen at 303K using 1.0g/L of the extract, while the lowest inhibition efficiency was shown at 333K using 0.2g/L of the extract, which was seen to be 12.91%. Increased extract concentration improved efficacy and surface coverage; increased temperature and corrodent concentrations worsened these qualities. Corrosion activation energy increased along with the rise in inhibitor concentration. The inhibited and uninhibited systems had activation energies of 50.127, 52.591, 53.574, and 54.066 KJ mol-1 and 39.879 KJ mol-1 in the 0.5M H2SO4 system, respectively. The pattern is also the same in 1.0M H2SO4, and the negative ΔGads values suggest that the adsorption process is doable. A non-spontaneous process is suggested by negative ΔSads values, whereas an endothermic process is suggested by positive ΔHads values. According to the examined FTIR spectrum, the Prosopis juliflora ethanol extract was discovered to include the functional groups (O-H, COOH, N-H, N=O, C-O). A scanning electron microscope (SEM) analysis provided evidence of the extract's ability to preserve the metal surface. However, the Freundlch adsorption isotherm was best suited to handle the adsorption data. According to the results, the data from Gads indicated that the extract inhibition process was physisorption, which is why the values were less than 40 KJ mol-1. According to the study's findings, a different concentration of the corrodent should be used to measure the extract's level of inhibition on the surface of mild steel.
Graphical Abstract
Keywords
Main Subjects
Introduction
It is believed that corrosion is a ubiquitous process that happens practically everywhere, including in the soil, water, and other environments we come into contact with. Rust is the common name for it [1]. It is an intolerable phenomenon that shortens the lifespan of the materials and ruins their attractiveness of metals [2–3]. Compared to other conditions, metallic components exposed to acidic media are more corrosion-resistant. Metallic materials are exposed to acidic environments in the oil and gas industry during chemical cleaning, pickling, and rescaling of most tanks with corrosive reagents like acid and other mediums that corrode the metals [1-3]. Corrosion inhibitors must be applied to either stop or eliminate the corrosion process. Anodic and cathodic protection, lubrication, painting, and electroplating are some methods utilized to combat corrosion-related issues [4-5]. But using a corrosion inhibitor is the best option. Although poisonous and nonbiodegradable inorganic inhibitors were used to suppress toxic metals [6-7]. Studies are now focused on helping metals from nonbiodegradable inhibitors. Plant extracts are considered a rich source of different phytochemical substances, including utilizing organic molecules with unsaturated bonds and hetero-polar atoms like S, N, O, and P [8], which are more biodegradable. They adsorb to the metal surface, preventing the loss of electrons from the metal surface. Adsorption is one of the distinctive methods that inhibitors utilize to stop most corroding agents from acting on the surface of metals in various mediums [9]. Insofar as urgent action is required to stop metal deterioration, the metallic world needs saving. A carbon-iron alloy called carbon steel has a maximum carbon concentration of 2.1 weight percent and a minimum defined number of other alloying elements. Low or mild-carbon steel, medium-carbon steel, and high-carbon steel are the three categories that carbon steel is divided into based on its carbon content [10]. The carbon steel most frequently used is low-carbon steel, sometimes referred to as mild steel. The metallic world requires rescue as immediate action is necessary to avert metal corrosion. Carbon steel is an iron-carbon alloy with a maximum carbon content of 2.1 weight percent and a minimum set of additional alloying elements. The three categories into which carbon steel is separated according to its carbon content are low or mild-carbon steel, medium-carbon steel, and high-carbon steel [10]. Low-carbon steel, commonly referred to as mild steel, is the form of carbon steel used most frequently.
Experimental
Mild Steel Coupon Preparation
At the Mechanical Engineering Department of Bayero University in Kano, the 0.36 cm thick iron sheets were press cut into 3 cm x 4 cm dimensions. The coupon was carefully scrubbed before use by smoothing the surface with silicon carbide emery paper of grades 120, 400, 800, and 1000. It was then thoroughly degreased in 100% ethanol, dipped in acetone, and dried in the open air [6-8].
Collection, preparation, and extraction of the plant samples
Fresh leaves of the plant Prosopis juliflora were obtained from Talatar jido Dawakin Kudu Local Government Area, Kano State, Nigeria. It was identified at the Department of Plant Biology of Bayero University Kano. The leaves were then dried, ground into powder form, and sieved to pass through a 250 nm mesh sieve. 200 g of the powdered sample was weighed into 1 L of absolute ethanol in an amber bottle. The bottle was properly corked and left to stand for two weeks with occasional shaking. The mixture was filtered and concentrated to free the extract from ethanol using a vacuum evaporator. The thick syrup was then air-dried for one week until a uniform mass was obtained.
0.2, 0.4, 0.6, 0.8, and 1.0 g/L of the extract in 0.5M and 1M H2SO4 were prepared by weighing 0.2, 0.4, 0.6, 0.8, and 1.0 g of the extract and dissolving each in 1litre of 0.5M and 1M H2SO4 respectively.
Blank corrodent solutions were also prepared by weighting 27.25 mL and 54.49 mL of the concentrated H2SO4 and made up to the mark with distilled water to prepare the 0.5M and 1M H2SO4 respectively, using the formula:
![]()
Where; Vc = Volume of concentrated acid to be diluted
M = Molarity of the dilute acid
VD= Volume of distilled water which dilution is to be made (Idm3=1000cm3)
Z= The molar mass of the substance
P= percentage assay of solute
S= specific gravity of the acids
Weight Loss Experiment
The pre-treated weighed coupons were individually and fully submerged in a 100 mL beaker half-fill with 0.5M and 1.0M of corrodent with and without the inhibitor after a tiny hole was drilled through one end of each coupon to make it hang in the solution and to be able to withdraw it quick from the test solution. The beakers were covered and put into a water bath with a thermostat at standard temperatures. The reaction was conducted for 1 h at 303, 313, 323, and 333K, respectively. The coupons were removed from the test solution, cleaned lightly with a soft brush and distilled water, dried in acetone, and reweighed. Three copies of the technique were completed, and the average was calculated. The weight loss experiment was conducted by varying temperatures (303, 313, 323, and 333K), corrodent concentration (0.5M and 1M H2SO4), and extract concentration (0.2, 0.4, 0.6, 0.8, and 1.0g/L) respectively.
The difference in weight was taken as the weight loss. From the weight loss values obtained, the corrosion rate (CR), inhibition efficiency (I.E), and surface coverage (θ) were calculated using equations (2–4), respectively [4-7].

Where W1 and W2 is the weight of the coupon before and after inhibition, respectively.
Where also θ is the degree of surface coverage
I.E is the inhibition efficiency (in %)
CR Corrosion rate ()
A is the area of the mild steel coupon (in Cm2) and t is the period of immersion (in hours)
Fourier Transform Infrared Analysis
The functional groups in the dried powdered leaves and ethanolic extract of Prosopis juliflora were identified using an FTIR instrument with model Carry 630 from Agilent Technologies. The mild steel coupons' corrosion product was measured using the same apparatus with and without an inhibitor. A wave number range of 4000-650 cm-1, 32 s, and 8 cm-1 resolution were used for the study [1-6].
Scanning Electron Microscopy
SEM was used to evaluate the surface morphology change brought on by contact with the test solutions and to track the impact of the inhibitor. After contacting 0.5M and 1M H2SO4 for an hour at 303K, mild steel coupon surfaces exposed to uncontrolled and inhibited corrosion systems underwent morphological investi-gations. This was done using a scanning electron microscope equipped with the PW-100-012 type and the 800-07334 DX7 Design Expert 716 software [12-15].
Electrochemical Studies
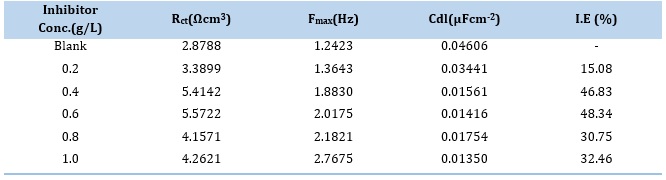
Electrochemical impedance spectroscopy (EIS) was carried out using AC potentiostat, manufactured by an ACM instrument equipped with EC-Lab software. The EIS studies were executed at the OCP of 10 kHz to 100 kHz as the frequency response at an amplitude of ac signal of 10mV. The testing setup consisted of three electrodes, a working electrode (Mild steel coupon) of 1cm2 area, a saturated calomel electrode as a reference electrode, and a platinum electrode as an auxiliary electrode. The corrosion behavior of the working electrode was investigated at a temperature of 303K; five concentrations of the corrosion inhibitor were used (0.2, 0.4, 0.6, 0.8 and 1.0 g/L) together with the blank of the 0.5M H2SO4. Nyquist plots were obtained from these experiments and used to analyze the impedance parameters such as charge transfer resistance (Rct), double layer capacitance (Cdl), and frequency (Fmax). The inhibitor efficiency was calculated from the Rct values using equation 5.

Where Rct(inh) and Rct represent the charge transfer resistances with and without inhibitors, respectively.
The Cdl was calculated using equation 6, [5-7].

Where Fmax is the frequency at which the impedance is maximum.
Results and discussion
Weight loss experiment
Using 0.5M and 1M H2SO4 at an elevated temperature, Prosopis juliflora was evaluated for corrosion inhibition in mild steel with corrodent and inhibitor concentrations ranging from 0.2-1 g/L. According to the information gathered and displayed in Table 1, weight loss reduced as the inhibitor's concentration increased from 0.2 g/L to 1g/L. The rate of corrosion decreases as the inhibitor concentration increases. The inhibitor's efficiency (IE) on the surface of the metal grows along with its concentration. The extract from Prosopis juliflora might explain this inhibitory efficiency leaves increasing as concentration increases its capacity to bind to the metal/electrolyte interface. The findings show that the inhibitor and corrodent concentrations are in good accord with the trend in corrosion rate. The experiment also showed a sizable change in the corrodent's concentration [8–9]. The system's capacity to inhibit growth decreases as the corrodant concentration increases from 0.5M to 1.0M H2SO4. This is seen in Table 1 as an increase in reaction rate as the corrodent concentration rises.
Table 1. Weight loss ΔW(g), Corrosion Rate CR(g/cm2h), Surface Coverage(Ө), Inhibition Efficiency(%I.E) of various concentrations of Prosopis juliflora in 0.5M and 1M H2SO4 solutions at varying Temperatures


Electrochemical impedance spectroscopy (EIS) measurements.
An important method for assessing the anticorrosion capabilities of metals is electrochemical impedance spectroscopy, which examines the impedance of the electrical double layer at the metal-medium interface. In a 0.5M H2SO4 solution, Figure 1 reveals the mild steel Nyquist plots at 300 °C in both the absence and presence of different inhibitor concentrations. The impedance response for mild steel in an unregulated acid solution increased when an inhibitor was added to the corrosive medium. It is also shown that the width of the capacitive loop rises as the inhibitor concentration increases and that the Nyquist plots take on the appearance of depressed semicircles, showing that the charge transfer mechanism controls the corrosion of mild steel. According to [10,11], the porous character of the inhibitor film formed on the metal surface and the surface roughness caused by inhibitor adsorption and corrosion product deposition are the causes of the Nyquist plot's depressed nature. Various impedance parameters such as charge transfer resistance (Rct), double layer capacitance (Cdl), and frequency (Fmax) were obtained from the Nyquist plot, and the inhibition efficiency (I.E) was calculated from these parameters as presented in Table 2.
The data show that the value of Rct increases with increasing inhibitor concentrations, indicating that corrosion inhibition on the metal surface increases at higher concentrations [12]. In contrast, the Cdl values decrease with increasing inhibitor concentrations due to the inhibitor molecules largely adsorbed on the mild steel-acid interface switching out water molecules [13]. This may mean that the local dielectric constant of the film decreases. This demonstrates that the inhibitory effectiveness shown in Table 2 was enhanced by the inhibitor's adsorption on the metal surface.
The %I.E obtained through the EIS methodology aligns with those gained utilizing weight-loss techniques. This result is in line with what was seen in [14–16].
 Figure 1. Nyquist plot for the corrosion of Mild steel in 0.5M H2SO4 at various concentrations of the extract
Figure 1. Nyquist plot for the corrosion of Mild steel in 0.5M H2SO4 at various concentrations of the extract
Fourier Transform Infra-Red Spectroscopy
Prosopis juliflora leaf extract in ethanol and corrosion products without and with inhibitors were subjected to FTIR spectroscopy. O-H stretching frequencies were identified from the FTIR spectra of the ethanolic extract of Prosopis juliflora (Figure 2) as being represented by a brief and faint band at 3696 cm-1. A large band seen at 3204 cm-1 is attributed to carboxyl acid, but a minor band seen at 2925 cm-1 was attributed to C-H stretching vibration. CC is responsible for the tiny wide band seen at 2125 cm-1. The strong absorption peak at 1607 cm-1 is caused by the N-H bending vibration of primary and secondary amines and amides, whereas the N=O causes the peak at 1514 cm-1 from an aromatic ring. At 1447 cm-1, 1346 cm-1, 1205 cm-1, and 1033 cm-1, respectively, stretch absorption is attributed to C=C from aromatic rings, C-O stretch, alkyl amine, and C-O vibration in alcohol. The absorption peaks below 1000 cm-1 are predominantly caused by the aliphatic and aromatic C-H functional groups. According to the FTIR spectrum, the Prosopis juliflora extract comprises O and N atoms in aromatic rings and functional groups (O-H, COOH, N-H, N=O, C-O).
 Figure 2. FTIR spectrum of Prosopis juliflora before and after corrosion
Figure 2. FTIR spectrum of Prosopis juliflora before and after corrosion
Table 2. Impedance parameters obtained from the Nyquist plot of mild steel in 0.5M H2SO4 at various extract concentrations
Scanning Electron Microscopy
Coupons made of mild steel had their surface morphology photographed using a scanning electron microscope. Figure 3a and 3b displays surface images for the uninhibited (0.5 and 1M H2SO4) and inhibited at the minimum and highest concentrations of H2SO4 (0.2g/L and 1g/L). At 300 °C for an hour, the samples (3x4x0.36cm3) were immersed in the 0.5M and 1M H2SO4 solutions. According to the findings, some scratches can be attributed to uneven surface polishing during preparation in the mild steel's surface morphology image prior to immersion in acid solution. The uninhibited systems shown in Figure 3 had surface cracks, spots, and pits, with the 0.5M H2SO4 system being more severely damaged than the 1M H2SO4 system. The metal displays a noticeable shift in morphology when 1 g/L of the inhibitor is present in both the 0.5M and 1M H2SO4 solutions, and its surface morphology is considerably less degraded in inhibited acid solutions than it is in unrestricted acid solutions. Accordingly, the justification implies that adding Prosopis juliflora leaf extract to acid solutions protected mild steel from experiencing severe corrosion by forming a shield on the metal's surface through the extract's adsorption.
 Figure 3a. SEM Micrographs of (A) mild steel in Blank 1M H2SO4 (B)
Figure 3a. SEM Micrographs of (A) mild steel in Blank 1M H2SO4 (B)
 Figure 3b. SEM Micrographs of (A) Mild steel in Blank solution of 0.5M H2SO4 (B) Mild Steel in Prosopis juliflora Solution of 0.5M H2SO4
Figure 3b. SEM Micrographs of (A) Mild steel in Blank solution of 0.5M H2SO4 (B) Mild Steel in Prosopis juliflora Solution of 0.5M H2SO4
Adsorption Isotherms
Adsorption isotherms provide vital information on the interactions between inhibitors and the correct active sites on the metal surface [16]. The surface coverage (θ) data show how Prosopis juliflora has covered the mild steel surface at various concentrations. Therefore, the Prosopis juliflora extract can slow down corrosion by either adsorbing plant materials to the metal surface or erecting barriers to keep the metal surface from coming into contact with the corrosive medium. The experimental data's fit to various adsorption isotherms supports this. This work examined the corrosion process using five isotherms: Langmuir, Temkin, Frumkin, Flory-Huggins, and Fruendlich. It was found that Freundlich gave the best depiction of inhibitor adsorption by considering that the Freundlich isotherm depicts physical adsorption across species and that its R2 values are close to unity, as shown by the equation.
![]()
Where, θ is the degree of surface overage, Cinh is the concentration of the inhibitor, Kads is the adsorption equilibrium constant, and the value of 1/n reveals the adsorption easiness of the inhibitor molecules on the surface of the corrosion-vulnerable material [6-13].
 Figure 4a. Freundlich isotherm for the adsorption of ethanolic extract of Prosopis juliflora on the surface of Mild steel in 0.5M H2SO4
Figure 4a. Freundlich isotherm for the adsorption of ethanolic extract of Prosopis juliflora on the surface of Mild steel in 0.5M H2SO4
 Figure 4b. Freundlich isotherm for the adsorption of ethanolic extract of Prosopis juliflora on the surface of Mild steel in 1.0M H2SO4
Figure 4b. Freundlich isotherm for the adsorption of ethanolic extract of Prosopis juliflora on the surface of Mild steel in 1.0M H2SO4
Table 3. Adsorption Parameters for the Adsorption of Prosopis juliflora on mild steel surface

Thermodynamic Studies
Free Energy of Adsorption
According to Equation 8, the standard free energy of adsorption, ΔGads, is linked to the Kads derived by extrapolating the straight line obtained from the Freundlich isotherm.
![]()
Where ΔG0ads is the change in the Gibbs free energy of adsorption, Kads is the equilibrium constant for adsorption determined from the intercept of each adsorption isotherm, R is the universal gas constant, T is the system's temperature, and 55.5 molL-1 is used as the unit of measure for the amount of water in a solution.
As a rule, a ΔG0ads value up to -20 KJ/mol denotes physisorption, a kind of adsorption that involves the electrostatic interaction of charged molecules with surfaces of charged metals. ΔG0ads values greater than -40 kJ/mol, on the other hand, point to chemisorption, the process of charge transfer or sharing from inhibitor molecules to the metal surface. Based on the results, all of the tested isotherms' computed ΔG0ads values in Table 4 are negative and less than the -40 kj/mol cutoff. The Freundlich isotherm in 0.5M H2SO4 had the highest ΔG0ads value of -26.8497kj/mol, while the Flory-Huggin's isotherm had the lowest value at -5.1536 kj/mol. As a result, the adsorption of the Prospis juliflora leaf extract on mild steel is both possible and compatible with the physical adsorption process that involves inhibition brought on by electrostatic contact between the charged molecules and the charged metal.
Table 4. Energy parameters for the dissolution of Mild steel in H2SO4 in the absence and presence of different concentrations of Prosopis juliflora

Activation Energy
Equation 9 illustrates how the logarithm of the metal's corrosion rate (CR) in the H2SO4 solution was used to compute the thermodynamic functions for mild steel dissolving in the presence and absence of the inhibitor at different temperatures.
logCR = log A – Ea/2.303RT (9)
Where, CR is the corrosion rate of the metal, A is the Arrhenius pre-exponential factor, R is the universal gas constant, Ea is the activation energy, and T is the system's temperature.
The value of Ea was estimated from the Arrhenius plot of log CR against 1/T for the blank and inhibited solutions and is shown in Table 4 as a straight line with a slope equal to -Ea/2.303R. It was discovered that the calculated values of Ea were higher for the inhibited and lower for the uninhibited. With the addition of 0.2, 0.4, 0.6, 0.8, and 1.0 g/L of the inhibitor, the activation energy in 0.5M H2SO4 increases from 39.87942 kJ/mol for the blank to 50.12691, 50.7806, 52.59082, 53.57524, and 54.06649 kJ/mol, respectively. The 1M H2SO4 solution shown in Table 6 illustrates that this is likewise the situation. According to these findings, Prosopis juliflora reduces metal dissolution in media containing 0.5 and 1M H2SO4 [20–25]. A considerable decrease in the inhibitor's adsorption on the mild steel surface with rising temperature is the cause of the increases in activation energy.
Ea values >80 Kj/mol often denote chemical adsorption, whereas Ea values 80 Kj/mol generally denote physical adsorption. The results are shown in Table 4, where the Ea values are all under 80 Kj/mol, indicating a physical adsorption process.
Enthalpy and Entropy
The transition state equation was used to compute the activation enthalpy and entropy for the corrosion process.
![]()
Where, CR is the corrosion rate, h is the planck's constant, NA is Avogadro's number ΔHads the enthalpy of activation, and ΔSads the activation entropy.
The enthalpy and entropy of activation for the corrosion process were computed from the straight line with the slope equal to [Log(R/NAh)+ Sa/2.303R] and intercept equal to [Log(R/T) versus 1/T] and are shown in Table 4. From the results, it can be concluded that both the 0.5M and 1M H2SO4 Solutions have positive values of enthalpy that are all less than 80Kj/mol. However, the entropy of activation for the 0.5M and 1M H2SO4 solutions was negative. While mild steel is difficult to dissolve, the endothermic nature of the process is indicated by the positive value of enthalpy of activation in both the presence and absence of an inhibitor. In contrast, the negative value of entropy of activation indicates that the activated complex in the rate-determining step is a representation of association rather than dissociation, implying a reduction in disorderliness going from reactant to activate solutions.
Conclusions
The Prosopis juliflora leaves extract has a somewhat inhibiting effect on the corrosion of mild steel. Consequently, the corrosion process becomes more ambitious as extract concentration increases and becomes less aggressive when corrosion-causing agent concentration and temperature increase. According to the scanning electron microscopy (SEM) results, the surface of mild steel exhibits the potential to form a layer, and an analysis using Fourier transform infrared spectroscopy (FTIR) revealed that the adsorption of specific organic functional groups in the extract brings on the inhibition. Electrochemical impedance spectroscopy (EIS) research found that higher concentrations were related to stronger metal surface corrosion inhibition. The Freundlich adsorption isotherm was followed by adsorption thermodynamic experiments, demonstrating that the corrosion process was physical and endothermic. It is advised that different corrodent concentrations be used to determine the amount of extract inhibition on the mild steel surface based on the results of various studied methods.
Acknowledgment
I acknowledge my supervisor, Dr. Magaji Ladan of the Department of Pure and Industrial Chemistry at Bayero University Kano, for overseeing this research project.
Disclosure statement
The authors declare that they have no conflict of interest
Orcid
Safiyya Abubakar Minjibir : 0009-0006-4671-2670
Magaji Ladan : 0000-0002-5602-6483
HOW TO CITE THIS ARTICLE
Safiyya Abubakar Minjibir *, Magaji Ladan. Corrosion Inhibition Potential of Prosopis Juliflora Leaves Extract on Mild Steel in H2SO4 Solutions, Adv. J. Chem. A, 2023, 6(3), 311-323.
DOI: 10.22034/AJCA.2023.398774.1372


