Document Type : Original Research Article
Authors
1 Department of Chemistry, Ahmadu Bello University Zaria, Kaduna State, Nigeria
2 Department of Chemistry, Al-Qalam University Katsina, Katsina State, Nigeria
3 Department of Chemistry, Faculty of Physical Sciences, Federal University Dutsin-Ma, Katsina State, Nigeria
Abstract
Dolichos pachyrhizus (Annonaceae) has been traditionally used in Africa to treat syphilis and many other microbial infections. However, no phytochemical study and antimicrobial investigations have been conducted on this species. Thus there is need for the discovery and development of new antibiotics and anifungal. The triterpenoids [Lupeol (1) and β-amyrin (2)] isolated from rhizome of Dolichos pachyrhizus (Harm) were evaluated for their antimicrobial activity against some selected Gram positive bacterial, Gram negative bacterial and Fungal isolates. The antimicrobial activity of the crude extract and compounds isolated was determined using agar well diffusion method. The compounds exhibited antimicrobial activity against most of the tested bacteria with minimum inhibitory concentration (MIC) and minimum bactericidal/fungicidal concentrations (MBC/MFC) values ranging from 2.5 to 20 mg/ml and 5 to 40 mg/ml, respectively. The triterpenoids could be a potentially effective antimicrobial agent to combat infectious diseases. The structures of the compounds were elucidated using modern spectroscopic and spectrometric techniques such as nuclear magnetic resonance (NMR) and mass spectrometry (MS) to be Lupeol and β-amyrin.
Graphical Abstract
Keywords
Main Subjects
Introduction
Pathogenic microorganism resistance strains are becoming more prevalent and emerging on a daily basis [1]. In addition, antimicrobial drugs in one way or another are connected to side effects or toxicity [2]. Therefore, great improvements are needed to address these problems associated to the current chemotherapeutic agents [3].
Because they contain many components with therapeutic significance, plant extracts have been utilized for a wide range of medicinal purposes for many years. Based on the occurrence of specific health issues, these features differ from region to region. As they are known to contain potent pharmacological substances, such as those with antibacterial and antifungal action, medicinal plants have therefore received considerable attention [4-5]. Phytochemicals are known for protecting plants and humans against parasitic and microbial diseases [6].
Biological properties of phytochemicals such as antibacterial [2], antioxidant [7], anticancer [8], and anti-inflammatory activities [9-10] have been reported. Dolichos pachyrhizus Harms (D. pachyrhizus) Papilionaceae (Fabaceae) a branching, sub-erect and downing herb dispersed in both hemispheres tropics and a member of fabaceae family [3].
pachyrhizus Harms is a common curling climber, sprawling perennial with small trifoliate leaves, bearing narrow flat curving pods when mature that are 1.5-2.0 inches long and have a persistent style at the peak. It grows in rocky soil and is a leguminous plant. The pods have 5-6 ellipsoid, flattened seeds that are 1/8 to 1/4 inch long [11].
It originates from Mexico and Central America and is a tropical plant. Due to its delicious underground tubercles, D. pachyrhizus, sometimes known as "jicama," has been widely cultivated in Mexico since the pre-Columbian era [12]. It was first cultivated under the name "yam bean" throughout India, China, and the USA. Since ancient times, Nigeria has used more yam beans as culinary ingredients than any other nation [13-15].
These plants are used by local citizens as fish poison [16] and insecticide [17], while in customary medicine they are used against syphilis [18].
Several phytochemical and pharmacological studies have been reported on Dolichos species such as Dolichos mitis [19-22], Dolichos biflorus [23], Dolichos erosus [24-25], and Dolichos lablab [26] among others with potentially therapeutic and prophylactic importance. On the other hand, based on the literature, there is no any phytochemical and or pharmacological activity reported on the D. pachyrhizus specie. Therefore, the current study aimed to isolate, characterize, and assess the antimicrobial efficacy of the bioactive component(s) and crude ethyl acetate extract of D. pachyrhizus rhizomes.
Experimental
Materials and methods
Reagents and solvents
The following chemicals were used: diluted hydrochloric acid (HCl), potassium iodide (KI), concentrated sulphuric acid (H2SO4), sodium hydroxide (NaOH), 1 % ferric chloride (FeCl2), ethyl acetate (CH3COOC2H5), and methanol (CH3OH). The analytical-grade reagents and solvents used in this study were all procured from Sigma Aldrich in Germany.
Plant collection and identification
In February 2019, the Dolichos pachyrhizus rhizome was collected from Ajja bush in the Batsari Local Government Katsina State, Nigeria. The rhizome was identified at the Herbarium Unit, Department of Biological Sciences, Ahmadu Bello University Zaria, Kaduna State, Nigeria, with a specimen number (ABU09001) lodged.
General analysis
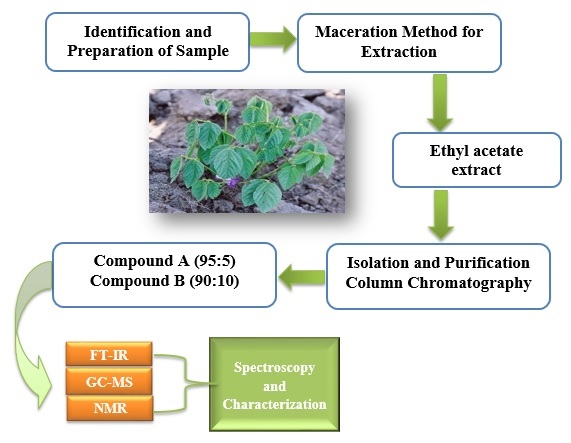
The extraction procedure used was the cold maceration approach as described by [27-28]. For the extraction, the following solvents in increasing polarity were employed to obtain crude extracts of D. pachyrhizus that were saved for further use:
n-Hexane, dichloromethane, ethyl acetate, and methanol. The compounds were separated from the ethyl acetate extract (12.5 g) using column chromatography (24 700 mm; Silica gel 60; 0.063 to 0.2 mm; 70 to 230 mesh ASTM; Merck, Darmstadt, Germany).
The mobile phase was a hexane-ethyl acetate solvent gradient system (90:10 to 30:70) and fractions of 10 mL were collected. Four mixed fractions were obtained from the collected fractions after thin layer chromatography (TLC) analysis using silica gel 60 TLC aluminum sheets (20 cm 20 cm, F254). Compounds 1 (0.72 g) and 2 (1.14 g) were obtained after additional separate purification with hexane and ethyl acetate of the second and seventh mixed fractions. The melting point was determined using melting point equipment (Shalom Instruments Supplies, Durban, R.S.A.).
Nuclear magnetic resonance (NMR) techniques (1H, 13C, and DEPT) (in CDCl3, Bruker 600 MHz) and infrared (FTIR) (KBr, Perkin-Elmer 100 FTIR) techniques were used to establish and confirmed the structures of the triterpenoids where chemical shifts (δ) were expressed in (ppm).
 Scheme 1. Structure of Lupeol (1) and β-amyrin (2)
Scheme 1. Structure of Lupeol (1) and β-amyrin (2)
Phytochemical Screening
To identify the existence of bioactive components, a phytochemical study of the ethyl acetate extract of D. pachyrhizus was carried out [29-30].
Column chromatographic purification
Crude ethyl acetate extract (2 g) was weighed and transferred into a packed column carefully on top of silica gel; about 5 g of dried silica gel was applied to serve as a protective layer. A gradient solvent system of 100 % n-hexane (100 cm3) was initially used to elute the column, followed by n-hexane-ethyl acetate (100 cm3) in this order; 95:5; 90:10; 85:15, 80:20, 75:25, 70:30, 65:35, 60:40, 55:45; 50:50; 45:55; 40:60; 35:65; 30:70; 25:75; 20:80; 15:85, and 10:90; 5:95.
Finally, the column was cleansed with 100% ethyl acetate (100 cm3). Different fractions were collected in a 10 cm3 beakers labeled (1-114) at intervals. At room temperature, each fraction was allowed to evaporate before examined through thin layer chromatography (TLC). Similar fractions were merged together based to their TLC pattern, grouped into (K, L, M, and N), and then purified further by a short column [23], using n-Hexane: Ethyl acetate (95:5 and 90:10) as solvent systems. This led to isolation of two compounds from fractions A and B which were thoroughly washed with pentane to obtain optimal purity for spectroscopic analyses labeled isolates 1 and 2.
Antimicrobial activity
Experimental organisms
The following species of bacteria were used to determine the antibacterial activity; four Gram-positive Staphylococcus aureus (S. aureus), (Bacillus cereus (B. cereus), Streptococcus pneumonia (S. pneumonia), and Staphylococcus saprophyticum (S. saprophyticum)) and four Gram-negative (Escherichia coli (E. coli), Klebsiella pneumonia (K. pneumonia), Selmonella typhi (S. typhi), Pseudomonas aeruginosa (P. aeruginosa)), Candida albicans (C. albicans), Chrysosporium merdarium (C. merdarium), and Trichophyton rubrum. (T. rubrum).
Determination of Zone of Inhibition
According to [31], the agar well diffusion method was employed. The ethyl acetate extract and isolated components from the Dolichos pachyrhizus rhizome were tested for their antibacterial activity. With the help of a sterile stick, the standardized innocula of the extract and isolated chemicals were uniformly smeared onto freshly prepared Mueller Hinton agar plates.
Each agar plate had five properly labeled wells drilled through it using a sterile cork borer (8 mm in diameter).
Each well received 0.2 mL of the proper concentration of extract and isolated compound, which was then given time to disperse into the agar. The inocula isolate was streaked onto a second plate, which was then covered with ciprofloxacin standard (10 ug/disc). The plates were incubated for 24 hours at 37 °C for the bacteria, and 48 hours at 25 °C for the fungi using Sabouraud Dextrose Broth. The plant extract and isolated compounds' antibacterial properties were measured in terms of the diameter of the inhibitory zones they formed.
Determination of Minimum Inhibitory Concentration (MIC)
The broth dilution procedure described by [32] was used to determine the lowest inhibitory concentration of the extract and isolated compounds. Mueller Hinton broth (MHB) was placed in test tubes along with various amounts of the extract and isolated compounds that shown antibacterial action against the test organisms. Each tube containing the diluted extracts received an inoculation of the organisms. For bacteria, the tubes were incubated at 37 °C for 24 hours, and for fungus, at 25 °C for 72 hours. The minimum inhibitory concentration (MIC) was defined as the lowest concentration in the series at which the test organisms showed no discernible growth.
Minimum bactericidal/fungicidal concentration (MBC/MFC)
By analyzing the test tube contents in the MIC estimations, the minimum bactericidal/fungicidal concentration was established. A loopful of each tube's contents was streaked onto a solidified nutrient agar plate as an inoculum, after which it was incubated at 37 °C for 24 hours to grow bacteria and at 25 °C for 48 hours to grow fungus. The minimum bactericidal/fungicidal concentration was determined to be the subculture's lowest concentration when there was no growth [32].
Results
Preliminary phytochemical test
Table 1. Preliminary phytochemical constituents of D. pachyrhizus extract

Electrical properties of SrSe/ZrSe deposited at a different time interval
Spectroscopic analyses
Isolated compound 1 was eluted with 95 % n-hexane: ethyl acetate and obtained as white sparkles (12 mg) which was examined for the chemical formula C30H50O by EIMS m/z 426.40 [M+]. Melting point ranges 210 - 212 °C.
In the IR scale, a very intensely wide peak was observed at 3325.04 cm-1 (OH Stretch), 2941.90 cm-1 (CH2, CH3 Stretch), 1638.02 cm-1 (C=C Asymmetric stretch), 1035.49 cm-1 (C-O Stretch of 2o alcohol), and 887.81 cm-1 (=C-H bend external CH2). Analysis of 1H-NMR (400 MHz, DMSO, δ, ppm): 4.65 (2H, s, H-29b), 4.57 (2H, s, H-29a), 3.07-3.10 (1H, m, H-3), 2.95-3.00 (1H, m, H-19), 2.35-2.38 (2H, t, J = 4.08 Hz, H-15), 1.83–1.91 (2H, m, H-21), 1.67 (3H, s, H-30), 1.50–1.62 (8H, m, H-1,2,6,13,18), 1.23–1.37 (7H, m, H-7,9,16,22), 1.06–1.17 (4H, m, H-11,12), 1.02 (3H, s, H-27), 0.95 (3H, s, H-26), 0.92 (3H, s, H-25), 0.81 (3H, s, H-24), 0.77 (3H, s, H-23), 0.75 (3H, s, H-28), and 0.61-0.62 (1H, t, J = 3.52 Hz, H-5). 13C-NMR (400 MHz, CDCl3) δ (ppm): 150.9, 108.5, 78.3, 55.5, 50.6, 49.2, 48.0, 43.0, 42.2, 40.5, 40.0, 38.7, 38.5, 38.2, 37.0, 36.9, 34.2, 30.4, 29.5, 29.3, 27.2, 25.5, 22.8, 20.7, 18.2, 18.0, 16.0, 15.3, 14.7 and 13.7 for carbon-20, 29, 3, 5, 9, 18, 19, 17, 14, 8, 22, 4, 1, 13, 10, 16, 7, 21, 23, 2, 15, 12, 11, 30, 6, 28, 25, 26, 24, and 27, respectively.
Isolated compound 2 was eluted with 90 % n-hexane and obtained as white shapeless solid (10 mg) which was examined for a chemical formula C30H50O via EIMS m/z 426 [M+]. Melting point was determined to be 212 - 214 °C. In the IR spectrum, an extremely prominent peak at 3488.30 cm-1 for hydrogen bonded, 2933.50 cm-1 for carbon-hydrogen stretch in CH2 and CH3, 1643.40 cm-1 for C = C asymmetric stretch and 1035.50 cm-1 for C-O stretch for 2o alcohols were observed. 1H-NMR (400 MHz, CDCl3): δ 3.15 (H-3), 0.67 (H-5), 5.22 (H-12), 0.77 (s, H3-23), 0.90 (s, H3-24), 0.74 (s, H3-25), 0.94 (s, H3-26), 1.16 (s, H3-27), 1.06 (s, H3-28), 0.86 (s, H3-29), 0.79 (s, H3-30) 5.22 (1H, br s, H-12), 3.61 (1H, dd, J = 8, 4.4 Hz, H-3), 0.80 (3H, s), 0.84 (3H, s), 0.88 (3H, s), 0.95 (3H, s), 1.12 (3H, s), 1.34 (3H, s) , 1.01 (3H, s), and 1.11 (3H, s). 13C-NMR (400 MHZ, CDCl3): δ (ppm); 158.1, 116.9, 79.1, 55.5, 49.3, 48.8, 41.3, 39.0, 38.8, 38.0, 37.7, 37.7, 37.6, 36.7, 35.8, 35.1, 33.7, 33.4, 33.1, 29.9, 29.8, 28.8, 28.0, 27.2, 25.9, 21.3, 18.8, 17.5, 15.5, and 15.4 for Carbon 13, 12, 3, 5, 9, 18, 19, 14, 8, 4, 1, 10, 22, 21, 7, 17, 20, 16, 2, 29, 30, 28, 15, 27, 11, 23, 24, 6, 26, and 25, respectively.
Antimicrobial screening
Table 2. Zone of inhibition of crude ethyl acetate extract and isolated compounds from the rhizome of D. pachyrhizus against tested pathogens in millimeter (mm)
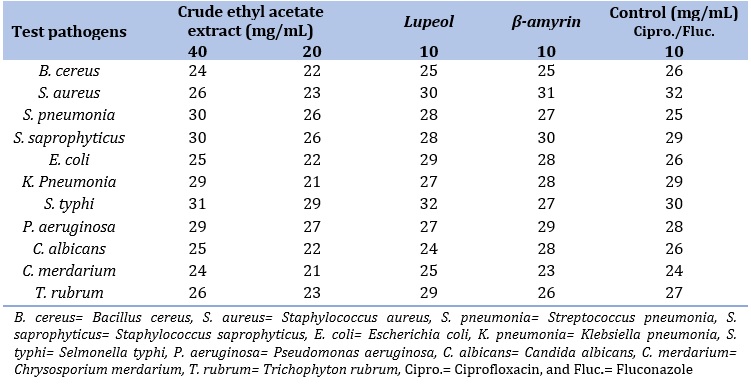
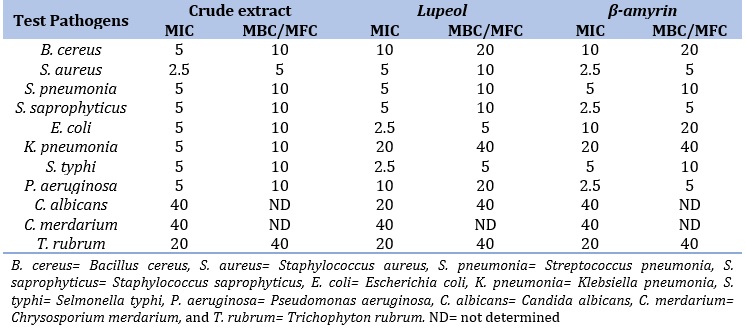
Table 3. Minimum Inhibition Concentration (MIC) and Minimum Bactericidal/Fungicidal Concentrations (MBC/MFC) for crude extract and isolated compounds against tested microorganisms in mg/mL
Discussion
The rhizome of Dolichos pachyrhizus indicates that anthraquinones, flavonoids, steroids, and terpenoids from ethyl acetate extract are present (Table 1). Plants with biological activity are frequently discovered to contain phytochemicals, which are compounds with biological action [33]. The findings suggest that the rhizome contains a substantial amount of secondary metabolites, including terpenoids with extreme medical significance that has been shown to support anti-diuretic activity [34]. In addition, they comprise the majority of the most beneficial drugs for treating human physiology, including those with anti-plasmodial, analgesic, antispasmodic, anti-diabetic, and anti-inflammatory properties [35,36].
The most diverse classes of phenolic compounds found in plants are flavonoids, which have antibacterial, anti-inflammatory, anti-allergic, and tumor-fighting effects [37-39]. Free radical scavengers, super antioxidants, and potentially water soluble flavonoids protect cells from oxidative damage. The presence of flavonoids, terpenoids, and steroids were confirmed via phytochemical tests of plant’s specie (Dolichos lablab seed extracts) [40-41]. From methanolic extract of the rhizome of the plant, flavonoids, terpenoids, and steroids were also confirmed [3]. Steroids have been shown to have potent anti-cancer, anti-allergic, antithrombotic, vasoprotective, tumor inhibitory, and antiviral properties [42-44].
Triterpenoid glycosides, also known as steroids, have been reported to have anti-inflammatory, cardiac depressant, and hypocholesterolemic effects [45]. The presence of steroids was confirmed; steroidal chemicals are significant and fascinating in pharmacy because of their interactions with other drugs, such as sex hormones, for which the steroidal structure may act as a potent precursor [43]. Dolichos lablab also exhibited antidiabetic, anti-inflammatory, analgesic, cytotoxic, antioxidant, hypolipidemic, insecticidal, antimicrobial, antilithiatic, hepatoprotective, and antispasmodic properties, according to the preliminary pharmacological tests [46].
To maintain their hormonal stability, nursing women or expectant mothers may find a lot of benefit from the decoction of the Dolichos pachyrhizus rhizome [47].
The findings of the phytochemical screening are consistent with what [23] reported from the methanol extract of Dolichos biflorus Linn seeds, which suggests that the plant's rhizome may be used as an antibacterial agent.
A pentacyclic triterpene, which has a color and melting point similar to isolated compound 1, was produced as a white, colorless powder. It displayed a mass peak at m/z 426 [M]+ in the MS spectrum, which matched the chemical formula C30H50O. Isolated compound 1's positive Liebermann-Buchard test result indicates that the substance is a terpernoid. An extremely strong broad peak at 3325.03 cm-1, which is indicative of the O-H bond stretch, could be seen in the isolated compound 1's IR spectrum. There was also a noticeable band at 2941.86 cm-1 that is associated with an aliphatic C-H stretch. The unsaturated section of the C-H vibrations and a peak for the C-O stretch for secondary alcohol were observed at 1035.52 cm-1 and 1638.03 cm-1, respectively. The methylenic part's rocking motion was thought to be the cause of the fairly intense band at 632.
1H-NMR spectrum of isolated compound 1 showed seven CH3 singlets protons, two double bonded protons of methylene part and one oxygenated C-H proton at δH 1.68 (3H, br s, Me-30), 1.03 (3H, s, C-26), 0.97 (3H, s, C-27), 0.94 (3H, s, C-24), 0.83 (3H, s, C-25), 0.79 (3H, s, C-23), and 0.76 (3H, s, C-28); 4.57 (1H, dd, J = 1.2, 2.4 Hz, H-29a), 4.69 (1H, br d, J = 2.4 Hz, H-29b), and 3.19 (1H, d, J = 5.1, 10.6 Hz, H-3), respectively. The double bond between methylene carbon (C-29) and quaternary carbon (C-20) was supported by the methylene protons of H-29 while H-3 proton appears as triplet at 3.16 ppm.
On the other hand, 13C-NMR spectrum of the isolated compound 1 shown the presence of thirty (30) carbon, further classified into seven (7) CH3, eleven (11) CH2, six (6) methines, and six (6) quaternary carbon atoms at C-23, C-24, C-25, C-26, C-27, C-28, and C-30; C-1, C-2, C-6, C-7, C-11, C-12, C-15, C-16, C-21, C-22, and C-29; C-3, C-5, C-9, C-13, C-18, C-19, and C-4, C-8, C-10, C-14, C-17, and C-20 together with one external methylene and oxygenated carbons at δC 108.52 and 78.27, respectively. The chemical shift values (in ppm) of 13C-NMR spectrum were obtained and compared with available data from literature (Table 4).
Table 4. 13C Spectroscopic data values of isolated compound 1 compared with literature values (400 MHz, CDCl3)

The bonding structure relationship of the compound 1 was confirmed via the 1H and 13C spectra of long range correlation. The correlation of proton at δ 1.68 (H-30) with quaternary carbon at δ 150.96 (C-20), CH2 carbon at δ 108.52 (C-29), and CH carbon at δ 47.99 (C-19) showed that the CH3 carbon atom (C-30) binds to the quaternary carbon (C-20). The oxygenated carbon with proton H-3 and the de-shielded signal at 78.27 are connected by methylene proton H-2 at 1.65 ppm [8,48].
Comparing the data from the literature with the one from 1H-NMR, 13C-NMR, and DEPT spectra revealed that compound 1’s structure is a lupeol (Scheme 1). Isolated compound 2 was also obtained as a white residue and its mass spectroscopic information projected that it had the same chemical formula as isolated compound 1 (C30H50O), that was confirmed via 13C-NMR spectrum. Isolated compound 2 displayed a positive test for terpenoids, just like isolated compound 1 above. The isolated compound 2's IR spectrum had an extremely broad peak at 3488.29 cm-1, which is usual of the O-H bond stretch. An aliphatic C-H stretch can be assigned by a reasonably strong band at 2933.48 cm-1. An intense band of C=C vibrations was seen at 1643.43 cm-1, and the comparable C-H vibrations of the unsaturated region were seen at 1013.8 cm-1.
A proton signifying the H-3 of a terpene moiety that emerged as a triplet at 3.26 and a different proton at 5.18 that suggested the presence of a tri-substituted olefinic bond were observable in the 1H-NMR spectrum of the isolated compound 2.
13C-NMR spectrum of the isolated compound 2 revealed to consist of thirty (30) carbon atoms which were further classified to eight (8) CH3, ten (10) CH2, five (5) CH, and seven (7) quaternary carbon atoms at C-23, C-24, C-25, C-26, C-27, C-28, C-29, and C-30; C-1, C-2, C-6, C-7, C-11, C-15, C-16, C-19, C-21, and C-22; C-3, C-5, C-9, C-12, and C-18, and also C-4, C-8, C-10, C-13, C-14, C-17, and C-20, respectively. These carbon signals were measured in ppm and compared to data from the literature (Table 5).
Table 5. 13C spectroscopic data of isolate 2 compared with literature (400 MHz, CDCl3)
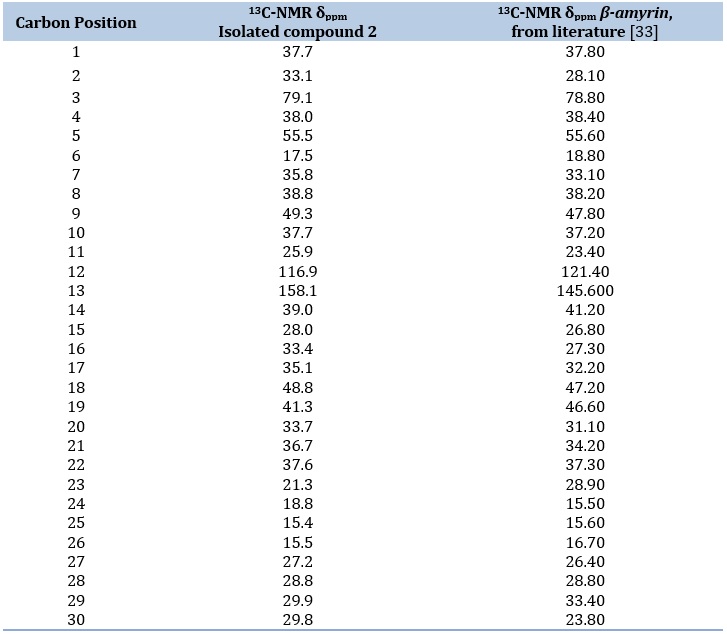
1H- and 13C-NMR chemical shift values were assigned to all the protons and carbons based on spectroscopic information. A literature search revealed that isolated compound 2's spectral data supported the structure of an oleanane triterpenoid framework (β-amyrin) with an OH at the C-3 position and an oleifinic bond at C-12 and C-13 position [33], this was additionally confirmed via a crucial COSY and HMBC correlations as presented in Figure S12. Consequently, structure of isolated compound 2 (Scheme 1) was found to be in line with information found in the previously reported work [33]. Triterpenoids (pentaccyclic triterpenes) with thirty (30) carbons skeleton, represent a varied class of natural products which are produced by arrangement of squalene epoxide molecules [35,49-50]. The triterpenoids, lupeol, and β-amyrin are powerful bioactive [10,33,51] and strong insecticidal and acaricidal compounds [20,30,33] found in diverse medicinal plants. Lupeol and β-amyrin have a wide range of bioactivities and bioassays that have been studied [52], suggesting that they have beneficial therapeutic characteristics with a variety of actions against different diseases. These compounds were reported to be acaricidal [20], antiangiogenic [53], antioxidative [38,54], and anti-inflammatory in nature [51,55]. They inhibit early stages of benzoyl peroxide-induced tumor development [56]. They are also crucial for normalizing the lipid profile [45], the activity of wound healing [36], the preventive impact against hypercholesterolemia linked to kidney damage, and the suppression of immunological factors [49].
In vitro antimicrobial activity
Antimicrobial activity (in vitro) of crude ethyl acetate rhizome’s extract and isolated compounds of D. pachyrhizus was determined by the use of agar well diffusion method against eight pathogenic strains of bacteria: Bacillus cereus, Staphylococcus aureus, Streptococcus pneumonia, and Staphylococcus saprophyticum (Gram positive bacteria), Escherichia. coli, Selmonella. Typhi, Klebsiella pneumonia, and Pseudomonas aeruginosa (Gram negative bacteria), and Candida albicans, Chrysosporium merdarium, and Trichophyton rubrum (Fungal isolates), and also compared to Ciprofloxacin and Flucunazole as commercial antibiotic and antifungal.
The biological activity of the test extract and isolated compounds was expressed in clear inhibition zone (mm) at various concentrations of the crude extract. The inhibition zone of the extract increases with increasing concentration (20 and 40 mg/mL) against the tested microbes. The results in Table 2 revealed that isolated compound 1 (Lupeol) showed highest inhibition zone against E. coli, S. typhi, C. merdarium, and T. rubrum 29, 32, 25, and 29 mm, respectively. These higher zones of inhibition recorded from lupeol were in agreement with what was reported by Babii et al. [50] which also agrees with the herbal use of the plant rhizome in the management of typhoid, diarrhea, and urinary tract infections. These values were high than the zone of inhibitions recorded from the crude ethyl acetate extract, β-amyrin, and the standard used antibiotic drug. β-amyrin, on the other hand, was found to be highly active against P. aeruginosa and Candida albicans with the highest inhibition zone of 29 and 28 mm, respectively, compared to crude extract, lupeol, and ciprofloxacin. These high zones of inhibition corroborate with the zones of inhibition reported from Neorautanenia mitis aqueous extract [51] and lower than the values reported from crude methanolic extract of Dolichos erosus [52,53]. These are also in line with the local use of the rhizome in managing pneumonia, urinary tract, and vaginal infections [54]. Same activity index (30 mm) was recorded from crude ethyl acetate and β-amyrin isolated from the plant against S. saprophyticus which were all greater than the zone of inhibition recorded from the standard antibiotic (29 mm) used and also agrees with the local use of the rhizome in the treatment of vaginal infections in women and urethritis in herbal traditional practice [54]. Similar zones of inhibition with standard drug were determined from the isolated compounds (25, 25, and 26 mm) against S. aureus, (30, 31, and 32 mm) against S. aureus and (27, 28, and 29) against K. pneumonia for lupeol, β-amyrin, and ciprofloxacin, respectively. These values prove active potentials of the isolated compounds in developing new antibiotic for treating pneumonia, meningitis, vomiting, and diarrheal syndrome as well as dermal and epidermal tissues problems [55]. The crude ethyl acetate extract shows the highest inhibition zone (30 mm) against S. pneumonia. The value is greater than the value recorded for the two isolated compounds and the standard drug (28, 27, and 25 mm) lupeol, β-amyrin, and ciprofloxacin, respectively, and showed synergistic potential of the combination therapy of the compounds.
MIC which is the lowest concentration of a substance or drug that inhibit growth of microorganism and MBC/MFC as a minimum concentration of a substance or drug that completely kills microorganisms are presented in Table 3. The MIC and MBC/MFC values of the crude extract and isolated compounds against test microorganisms ranged from 2.5-40 mg/mL. Least MIC values (2.5 mg/mL) obtained showed potential property of the crude extract and compounds compared to the antibiotic drug used as control against S. aureus, S. saprophyticus, E. coli, S. typhi, and P. aeruginosa which also correspond to the high zones of inhibition against the microbes and ethnomediinal importance of the plant [56]. The high MIC and MBC/MFC recorded revealed moderate to low active the drugs are and determined their lower zones of inhibitions.
The highest zones of inhibition (24-32 mm), at 10 mg/mL of the isolated compounds revealed full susceptibility against the tested bacterial strains. These high zones of inhibition recorded were higher than some reported inhibition zone against E. coli, S. saprophyticus, and S. typhi [31,51], lower than the values reported from Dolichos fiblorus [23], Dolichos mitis [43], and wood of Eucalyptus urograndis and Mimosa tenuiflora [34]. Values also recorded against S. pneumonia, S. saprophyticum, K. pneumonia, S. typhi, and P.aeruginosa (gram positive and negative bacterial strains) were greater than the values obtained from the positive control (ciprofloxacin) against the tested bacteria. These high activity observed from the study could possibly be associated with the use of the rhizome in the treatment of syphilis, cough, and typhoid in ethnomedicine. The isolated compounds, on the other hand, such as Lupeol and β-amyrin from the rhizome of the plant could be potential antibiotic agents susceptible against the bacteria and proved its folklore uses.
Conclusion
In conclusion, this study isolated and characterized two triterpenoid from ethyl acetate extract of Dolichos pachyrhizus rhizomes. The extract and isolated compounds exhibited significant activity to both test bacteria and fungi. These findings supported traditional use of D. pachyrhizus in treating various microbial infections. The bioactive compounds identified in this study hold promise for further exploration and development as potential antimicrobial agents. Future studies should focus on the purification, synthesis, and evaluation of these compounds for their therapeutic applications in the pharmaceutical industry.
Acknowledgment
We are grateful to all chemistry laboratory technologists from Ahmadu Bello University Zaria, Kaduna State, Federal University, Dutsin-Ma, Katsina State, and Al-Qalam University Katsina, Katsina State for their contribution in one way or the other during the research.
Disclosure statement
The authors declare that they have no conflict of interest.
Orcid
Nasiru Malan Musa : 0000-0002-4113-7228
Muhammad Sani Sallau : 0000-0002-0299-2467
Adebayo Ojo Oyewale : 0000-0003-2625-1636
Tijjani Ali : 0000-0002-8388-4686
HOW TO CITE THIS ARTICLE
Nasiru Malan Musa*, Muhammad Sani Sallau, Adebayo Ojo Oyewale, Tijjani Ali. Antimicrobial Activity of Lupeol and β-amyrin (Triterpenoids) Isolated from the Rhizome of Dolichos Pachyrhizus Harm. Adv. J. Chem. A, 2024, 7(1), 1-14.